- Siamenoside I
-
- $0.00 / 5mg
-
2023-02-24
- CAS:126105-12-2
- Min. Order: 5mg
- Purity: ≥98%(HPLC)
- Supply Ability: 10 g
- Siamenoside I
-

- $1.00 / 1kg
-
2019-07-06
- CAS:126105-12-2
- Min. Order: 1kg
- Purity: 95%-99%
- Supply Ability: 1000kg
|
| | Siamenoside I Basic information |
| Product Name: | Siamenoside I | | Synonyms: | SHIKONIN(SH);(3beta,9beta,10alpha,11alpha,24R)-3-(beta-D-Glucopyranosyloxy)-11,25-dihydroxy-9-methyl-19-norlanost-5-en-24-yl O-beta-D-glucopyranosyl-(1-2)-O-[beta-D-glucopyranosyl-(1-6)]-beta-D-glucopyranoside;Momordica grosvenori(Luo Han Guo );β-D-Glucopyranoside, (3β,9β,10α,11α,24R)-3-(β-D-glucopyranosyloxy)-11,25-dihydroxy-9-methyl-19-norlanost-5-en-24-yl O-β-D-glucopyranosyl-(1→2)-O-[β-D-glucopyranosyl-(1→6)]-;b-D-Glucopyranoside, (3b,9b,10a,11a,24R)-3-(b-D-glucopyranosyloxy)-11,25-dihydroxy-9-methyl-19-norlanost-5-en-24-ylO-b-D-glucopyranosyl-(12)-O-[b-D-glucopyranosyl-(16)]-;Inhibitor,Siamenoside I,inhibit;Semenin I/ Monk fruit I | | CAS: | 126105-12-2 | | MF: | C54H92O24 | | MW: | 1125.29 | | EINECS: | | | Product Categories: | chemical reagent;pharmaceutical intermediate;phytochemical;reference standards from Chinese medicinal herbs (TCM).;standardized herbal extract | | Mol File: | 126105-12-2.mol | 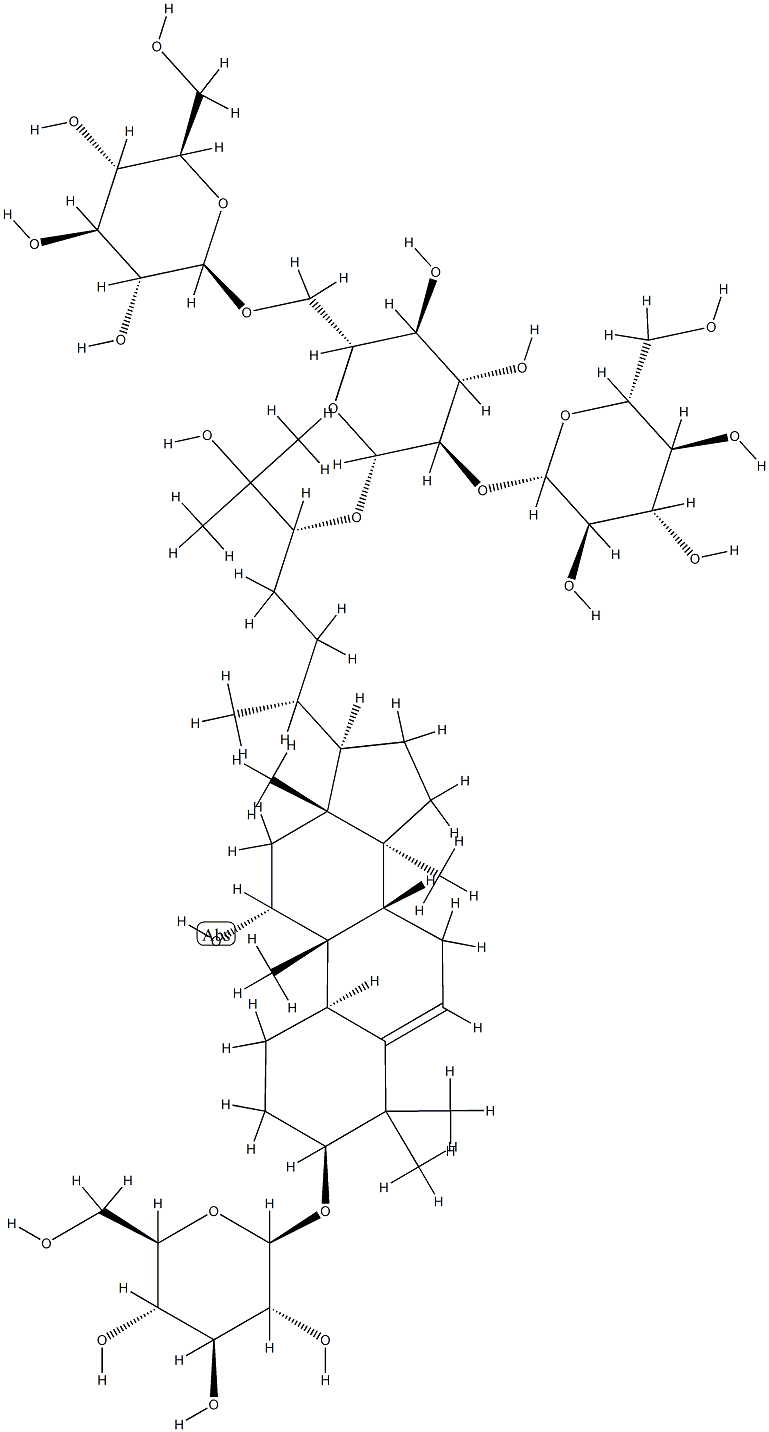 |
| | Siamenoside I Chemical Properties |
| Boiling point | 1179.3±65.0 °C(Predicted) | | density | 1.46 | | storage temp. | 2-8°C | | solubility | Ethanol: soluble | | form | A solid | | pka | 12.82±0.70(Predicted) | | color | White to off-white |
| | Siamenoside I Usage And Synthesis |
| Chemical Properties | White crystalline powder, soluble in water, insoluble in petroleum ether, dichloromethane, chloroform, benzene, ethyl acetate and other organic solvents, derived from Siraitia grosvenorii. | | Uses | Used for content determination/identification/pharmacological experiments, etc. | | Biological Activity | Siamenoside I is a mogroside and has an anti-cancer and antioxidant effect. | | in vivo | In rat, the metabolic reactions of siamenoside I include deglycosylation, hydroxylation, dehydrogenation, deoxygenation, isomerization, and glycosylation. Siamenoside I and its metabolites are mainly distributed to the intestines, stomach, kidneys, and brain. p> |
| | Siamenoside I Preparation Products And Raw materials |
|