|
|
| | CHLORINE MONOFLUORIDE Basic information |
| Product Name: | CHLORINE MONOFLUORIDE | | Synonyms: | Chlorofluoride;ClF;Fluorine chloride;Chlorine monofluoride 99%;Chlorinemonofluoride99%;fluoridochlorine;CHLORINE MONOFLUORIDE;Chlorine fluoride (ClF) (6CI,8CI,9CI) | | CAS: | 7790-89-8 | | MF: | ClF | | MW: | 54.45 | | EINECS: | 232-229-9 | | Product Categories: | | | Mol File: | 7790-89-8.mol | 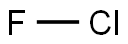 |
| | CHLORINE MONOFLUORIDE Chemical Properties |
| Melting point | -155,6°C | | Boiling point | -100°C | | density | 1,62 g/cm3 | | solubility | reacts with H2O | | form | colorless gas | | color | colorless | | Water Solubility | violent reaction with H2O [MER06] |
| | CHLORINE MONOFLUORIDE Usage And Synthesis |
| Chemical Properties | colorless gas; slightly yellow when liquid; enthalpy of vaporization 24 kJ/mol; specific conductivity 1.9×10?7 ohm· cm; destroys glass instantly, attacks quartz readily in presence of moisture; organic matter bursts into flame instantly on contact [MER06] [CRC10] | | Definition | ChEBI: Fluoridochlorine is a diatomic halohalide. | | Synthesis | A nickel or Monel cylinder within a furnace is used as a reaction vessel where chlorine and fluorine gases are introduced. The setup includes a condenser and traps to condense and separate the gases, with ClF being liquefied in a trap cooled by liquid nitrogen. The process involves heating to 400°C, adjusting the flow rate of chlorine for a given fluorine cell current, and collecting the product in a cooled trap. After fluorination, ClF is distilled into a steel cylinder, with the system pressure maintained at about 1 atm. The distillation stops when condensate accumulates in a quartz trap, and the yield is up to 90% based on chlorine. |
| | CHLORINE MONOFLUORIDE Preparation Products And Raw materials |
| Preparation Products | BROMINE TRIFLUORIDE-->1-Butanesulfonic acid, 1,1,2,2,3,3,4,4,4-nonafluoro-, anhydride with hypochlorous acid |
|