- Vonoprazan Fumarate
-

- $0.00 / 25kg
-
2024-03-28
- CAS:881681-01-2
- Min. Order: 25kg
- Purity: 98%
- Supply Ability: Inquiry
- Vonoprazan Fumarate
-

- $0.00 / 1g
-
2024-03-12
- CAS:881681-01-2
- Min. Order: 1g
- Purity: 98% HPLC
- Supply Ability: 1kg
|
| | Vonoprazan FuMarate Basic information |
| Product Name: | Vonoprazan FuMarate | | Synonyms: | Vonoprazan FuMarate###NA;1-(5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine fumarate;Vonoprazan-025-Salt1;Voranomazin;Vonoprazan FuMarate USP/EP/BP;Vonoprazan fumarate,TAK-438,Takecab;Vonoprazan fumarate 13C D3;Fumarate vonorazan | | CAS: | 881681-01-2 | | MF: | C21H20FN3O6S | | MW: | 461.46 | | EINECS: | 1592732-453-0 | | Product Categories: | | | Mol File: | 881681-01-2.mol | 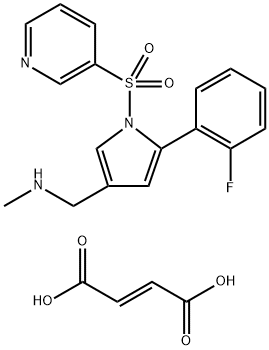 |
| | Vonoprazan FuMarate Chemical Properties |
| storage temp. | Store at -20°C | | solubility | DMSO: 50 mg/mL (108.35 mM) | | form | Solid | | color | White to Off-White | | InChIKey | ROGSHYHKHPCCJW-WLHGVMLRSA-N | | SMILES | C(/C(=O)O)=C\C(=O)O.S(C1=CN=CC=C1)(N1C=C(CNC)C=C1C1C=CC=CC=1F)(=O)=O |
| | Vonoprazan FuMarate Usage And Synthesis |
| Chemical Properties | Vonoprazan fumarate is white to nearly white crystals or crystalline powder which melts at 194.8°C . It can be dissolved in dimethyl sulfoxide, but has limited solubility
in N,N-dimethylacetamide, N,N-dimethylformamide, methanol, and water. It
is only slightly soluble in ethanol (99.5%), and has very low
solubility in 2-propanol, acetone, 1-octanol, and acetonitrile. | | Uses | Vonoprazan Fumarate (CAS# 881681-01-2) is a useful research chemical and a ATPase potassium-competitive acid blocker. | | benefits | Compared with traditional irreversible proton pump inhibitors (such as omeprazole, esomeprazole, etc.), Vonoprazan fumarate advantages:
① The onset of action is good, and the maximum acid-suppressing effect will be achieved on the first day of administration;
② Oral administration, not affected by gastric acid damage;
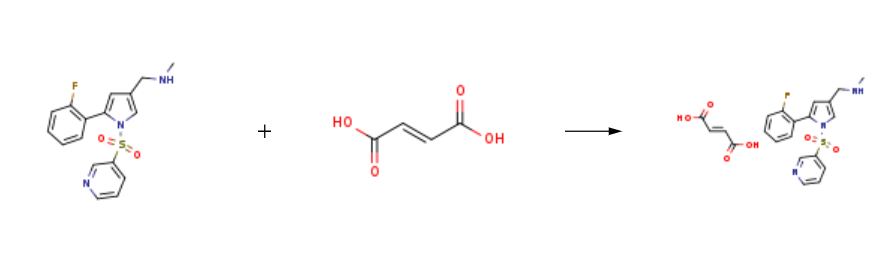
③It can improve the phenomenon of acid breakthrough at night. | | General Description | Vonoprazan fumarate (TAK-438) is a new oral anti-gastric acid drug jointly launched by Takeda Pharmaceutical and Otsuka Pharmaceutical. It can be used for the treatment of erosive esophagitis, gastric ulcer and duodenal ulcer. | | Mechanism of action | Vonoprazan fumarate is a potassium ion (K+) competitive acid blocker (P-CAB), which is a reversible proton pump inhibitor. It can inhibit the combination of K+ and H+-K+-ATPase (proton pump), so that secretion of gastric acid is terminated. Vonoprazan fumarate has a strong and lasting inhibitory effect on gastric acid secretion. | | Synthesis | Vonoprazan FuMarate is prepared by the reaction of vonoprazan and (2E)-but-2-enedioic acid. The steps are as follows:
Step 1: (2E)-but-2-enedioic acid With sodium hydroxide In water at 50 - 60℃.
Step 2: vonoprazan In water; toluene at 25 - 35℃; Reagent/catalyst.
Add 730g of water and 73g of fumaric acid to a 2L reaction flask at 5060, stir and disperse, control the temperature between 5060, add 25g of hydrogen hydroxide dropwise to the fumaric acid suspension The solution of sodium and 300 g of water was gradually dissolved in the system during the dropwise addition, and the aqueous solution of compound was obtained and kept for use.Between 25 and 35°C, take half of the toluene solution of compound obtained in the previous step and put it into a 5L reaction flask, and add the aqueous solution of the above compound dropwise to it. During the dropwise addition, the system has a white solid precipitation. After the dropwise addition was completed, keep stirring for 2-4 hours, filter, and dry the filter cake at 50°C to obtain 131.32 g of white solid, the HPLC purity was 99.96%, and the yield was 94%.
 |
| | Vonoprazan FuMarate Preparation Products And Raw materials |
|