|
|
| | 2-(3-bromothiophen-2-yl)ethanol Basic information |
| | 2-(3-bromothiophen-2-yl)ethanol Chemical Properties |
| Boiling point | 270.3±25.0 °C(Predicted) | | density | 1.651±0.06 g/cm3(Predicted) | | pka | 14.39±0.10(Predicted) | | InChI | InChI=1S/C6H7BrOS/c7-5-2-4-9-6(5)1-3-8/h2,4,8H,1,3H2 | | InChIKey | KCZAFTAJOFCEPQ-UHFFFAOYSA-N | | SMILES | C1(CCO)SC=CC=1Br |
| | 2-(3-bromothiophen-2-yl)ethanol Usage And Synthesis |
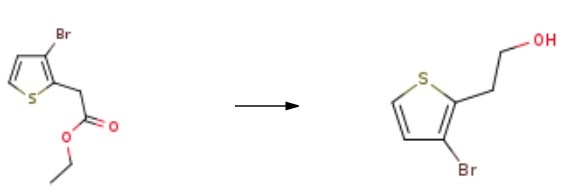
| Description | 2-(3-Bromothiophen-2-yl)ethanol, also known as 2-(3-bromothiophen-2-yl)ethanol, is an organic compound with the chemical formula C6H5BrSCH2CH2OH. It is a colorless liquid with a strong sulfur-like odor. It is used in various scientific research applications, including in the synthesis of pharmaceuticals, as a reagent in organic synthesis, and as a catalyst for cyclization reactions. It is also used as a corrosion inhibitor in the petroleum industry. | | Uses | 2-(3-Bromothiophen-2-yl)ethan-1-ol is a useful reagent in the copper halide catalyzed synthesis of alkyl aryl and alkyl heteroaryl ethers. | | Synthesis | A solution of ethyl 2-(3-bromothiophen-2-yl)acetate (4.40 g) in tetrahydrofuran (88.5 mL) was cooled to 0° C under a nitrogen gas atmosphere, lithium aluminum hydride (672 mg) was added to the solution, and the mixture was stirred for 0.6 h. Ice water, 1 mol/L hydrochloric acid, and ethyl acetate were added to the reaction mixture, and the resulting mixture was stirred. Then, the organic layer was successively washed with 1 mol/L hydrochloric acid, saturated aqueous sodium hydrogen carbonate, and saturated brine and dried. The solvent was evaporated under reduced pressure, and the resulting residue was purified by column chromatography (hexane/ethyl acetate) to obtain 2-(3-bromothiophen-2-yl)ethanol.
 |
| | 2-(3-bromothiophen-2-yl)ethanol Preparation Products And Raw materials |
|