- Iodomethyl pivalate
-

- $0.00 / 1KG
-
2024-09-18
- CAS:53064-79-2
- Min. Order: 1KG
- Purity: 98%min
- Supply Ability: 30tons/month
- Iodomethyl pivalate
-
- $8.00 / 1KG
-
2024-04-05
- CAS:53064-79-2
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
- Iodomethyl pivalate
-

- $7.00 / 1kg
-
2019-07-06
- CAS: 53064-79-2
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 100kg
|
| | Iodomethyl pivalate Basic information |
| Product Name: | Iodomethyl pivalate | | Synonyms: | AURORA KA-7367;IODOTOLUENE(4-);IODOMETHYL PIVALATE;(Pivaloyloxy)Methyl Iodide;2,2-DiMethyl-propanoic Acid IodoMethyl Ester;Iodomethyl Pivalate (stabilized with Copper chip);2,2-dimethylpropionic acid iodomethyl ester;iodomethyl 2,2-dimethylpropanoate | | CAS: | 53064-79-2 | | MF: | C6H11IO2 | | MW: | 242.05 | | EINECS: | 258-339-7 | | Product Categories: | Miscellaneous Reagents;Miscellaneous | | Mol File: | 53064-79-2.mol | 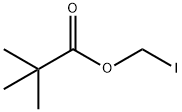 |
| | Iodomethyl pivalate Chemical Properties |
| Melting point | 33-35 °C(lit.) | | Boiling point | 211.5 °C(lit.) | | density | 1.614±0.06 g/cm3(Predicted) | | refractive index | 1.4830 to 1.4870 | | Fp | 194 °F | | storage temp. | Refrigerator, under inert atmosphere | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Oil | | color | Yellow | | Sensitive | Light, Air and Heat Sensitive | | InChI | InChI=1S/C6H11IO2/c1-6(2,3)5(8)9-4-7/h4H2,1-3H3 | | InChIKey | PELJISAVHGXLAL-UHFFFAOYSA-N | | SMILES | C(OCI)(=O)C(C)(C)C | | CAS DataBase Reference | 53064-79-2(CAS DataBase Reference) |
| RIDADR | UN 3265 8/PG III | | WGK Germany | 3 | | HS Code | 2915.90.5050 | | HazardClass | 8 | | PackingGroup | III |
| | Iodomethyl pivalate Usage And Synthesis |
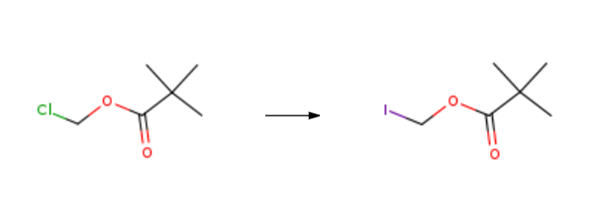
| Chemical Properties | Pale Yellow Liquid | | Uses | Iodomethyl pivalate can be used as reagent used for the addition of pivaloyl group. | | Synthesis Reference(s) | The Journal of Organic Chemistry, 48, p. 5280, 1983 DOI: 10.1021/jo00174a024 | | Synthesis | Iodomethyl pivalate is synthesised using chloromethyl pivalate as a raw material by chemical reaction. The specific synthesis steps are as follows:
Three bottles were added 10.0 g of chloromethyl pivalate, Ethyl acetate 30 mL, 11.6 g of sodium iodide and 3.6 g of calcium chloride, After heating to 78 ° C for 6 h, Cooling to 0 ,Then washed with 5% sodium thiosulfate to colorless, Dried over anhydrous magnesium sulfate, Concentrated under reduced pressure. 16 g of a yellow liquid. Iodomethyl pivalate was obtained. The reaction molar yield was 94% and GC: 98%.
 |
| | Iodomethyl pivalate Preparation Products And Raw materials |
|