- Ferrous sulfide
-
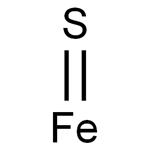
- $0.00 / 1KG
-
2024-09-04
- CAS:1317-37-9
- Min. Order: 1KG
- Purity: 99.0%
- Supply Ability: 10000KG
- Ferrous sulfide
-

- $6.00 / 1KG
-
2024-03-29
- CAS:1317-37-9
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
- Ferrous sulfide
-

- $1.00 / 1KG
-
2020-01-13
- CAS:1317-37-9
- Min. Order: 1KG
- Purity: 98%-99.9%
- Supply Ability: 200kg
|
| | Ferrous sulfide Chemical Properties |
| Melting point | 1195 °C | | Boiling point | decomposes [HAW93] | | density | 4.84 g/mL at 25 °C(lit.) | | vapor pressure | 0Pa at 25℃ | | storage temp. | no restrictions. | | solubility | insoluble in H2O; reacts with acid solutions | | form | Sticks | | color | Grayish-black | | Specific Gravity | 4.84 | | Odor | Odorless | | Water Solubility | Soluble in water(0.0062g/L ), in acids with evolution of hydrogen sulfide. Insoluble in nitric acid. | | Crystal Structure | NiAs type | | Sensitive | Moisture Sensitive | | Merck | 14,4058 | | crystal system | Six sides | | Solubility Product Constant (Ksp) | pKsp: 17.2 | | Space group | P63/mmc | | Lattice constant | | a/nm | b/nm | c/nm | α/o | β/o | γ/o | V/nm3 | | 0.345 | 0.345 | 0.567 | 90 | 90 | 120 | 0.0584 |
| | Stability: | Stable. Incompatible with strong acids, strong bases, metal oxides. Avoid moisture. | | CAS DataBase Reference | 1317-37-9(CAS DataBase Reference) | | EPA Substance Registry System | Ferrous sulfide (1317-37-9) |
| | Ferrous sulfide Usage And Synthesis |
| Uses | Iron(II) sulfide occurs in nature as the minerals magnetkies, troillite and pyrrhotine. The most important application of this compound is in Kipp’s apparatus as a source for laboratory preparation of hydrogen sulfide. It also is used in paints, pigments, and ceramics and lubricant coatings.
| | Preparation | Iron(II) sulfide may be synthesized from the elements but the product is contaminated with iron. The reaction is exothermic and the heat of reaction melts iron. Pure sulfide may be obtained by using a slight excess of sulfur: the excess then is distilled off.
The compound also may be precipitated by treating an aqueous solution of an alkali metal sulfide with that of iron(II) chloride or any iron(II) salt solution:
S2– (aq) + Fe2+ (aq) → FeS(s)
Another method of preparation involves passing a mixture of hydrogen sulfide and hydrogen over iron(III) oxide at about 1,000°C:
Fe2O3 + 2H2S + H2 → 2FeS + 3H2O
| | Reactions | Iron(II) sulfide reacts with acids evolving hydrogen sulfide:
FeS + 2HCl → H2S + FeCl2
The compound is readily oxidized under moist condition by action of air, forming triiron tetroxide and elemental sulfur:
3FeS + 2O2 → Fe3O4 + 3S
The above reaction is exothermic.
Iron(II) sulfide decomposes to its elements when heated above 1,100°C:
FeS → Fe + S
When heated with boiled water, it generates sulfuric acid and hydrogen:
4FeS + 8H2O + 7O2 → 4H2SO4 + 4H2 + Fe2O3
| | Chemical Properties | grey to brown-black lumps or powder | | Physical properties | Colorless hexagonal or tetragonal crystals; density 4.7g/cm3; melts at 1188°C; insoluble in water; soluble in acids (reacts). | | Uses | Iron(II) sulfide is used to generate hydrogen sulfide instantaneously by reacting with hydrochloric acid. In laboratory, Kipp?s apparatus is utilized for this purpose. It is used as a re-sulphurizing and alloying agent, as a reducing agent to remove heavy metal impurities from phosphoric acid. It is used to control hydrogen embrittlement in alloy and stainless steel industries. | | Uses | Ferrous sulfide can be used as a laboratory source of H2S; in the ceramic industry; as a paint pigment; in anodes; in lubricant coatings. | | Flammability and Explosibility | Non flammable |
| | Ferrous sulfide Preparation Products And Raw materials |
|