|
|
| | 5-Aminouridine Basic information |
| Product Name: | 5-Aminouridine | | Synonyms: | 5-aminouridine;5-AminouridineHClsalt;5-amino-1-(3,4-dihydroxy-5-methylol-tetrahydrofuran-2-yl)pyrimidine-2,4-quinone;5-amino-1-[3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]pyrimidine-2,4-dione;5-azanyl-1-[3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]pyrimidine-2,4-dione;5-Aminouridine Hcl;Uridine, 5-amino-(6CI,7CI,8CI,9CI);5-Amino-1-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione | | CAS: | 2149-76-0 | | MF: | C9H13N3O6 | | MW: | 259.22 | | EINECS: | | | Product Categories: | | | Mol File: | 2149-76-0.mol | 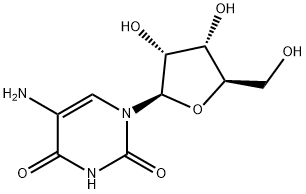 |
| | 5-Aminouridine Chemical Properties |
| Melting point | 214-216 °C | | density | 1.712±0.06 g/cm3(Predicted) | | storage temp. | −20°C | | pka | 9.31±0.10(Predicted) | | InChI | InChI=1S/C9H13N3O6/c10-3-1-12(9(17)11-7(3)16)8-6(15)5(14)4(2-13)18-8/h1,4-6,8,13-15H,2,10H2,(H,11,16,17)/t4-,5-,6-,8-/m1/s1 | | InChIKey | YBTWWWIJBCCYNR-UAKXSSHOSA-N | | SMILES | OC[C@H]1O[C@@H](N2C=C(N)C(=O)NC2=O)[C@H](O)[C@@H]1O |
| | 5-Aminouridine Usage And Synthesis |
| Description | 5-Aminouridine can modify nucleobases and can be incorporated into the target DNA. 5-Aminouridine exhibits a wide range of biological activity and it inhibits the growth of tumors, fungi and viruses.
| | in vitro |
This modified nucleobase, 5-Aminouridine, must exhibit a low enough potential to be oxidized by Os(bpy)33+. These modified nucleobases can be inserted into the target DNA by direct synthesis using modified phosphoramidites
| | in vivo |
5-Aminouridine inhibits the incorporation of carbamyl aspartate into pyrimidines of both RNA and DNA in rat lived and [32P]phosphate into phospholipids and RNA nucleotides of rat liver slices and hepatoma. 5-Aminouridine was found to be incorporated into the RNA of Ehrlich ascites cells, and 5-aminouridine monophosphate was shown to be a strong inhibitor of orotidine 5'-phosphate decarboxylase[1].
| | References | [1] David A. Smith, Donald W. Visser, P. Roy-Burman. “Studies on 5-aminouridine.” Biochimica et Biophysica Acta (BBA) - Nucleic Acids and Protein Synthesis 119 2 (1966): Pages 221-228. |
| | 5-Aminouridine Preparation Products And Raw materials |
|