- Zinc hydroxide
-

- $40.00 / 1kg
-
2024-02-29
- CAS:20427-58-1
- Min. Order: 1kg
- Purity: 0.99
- Supply Ability: 30 tons
- Zinc hydroxide
-

- $0.00 / 25KG
-
2023-09-20
- CAS:20427-58-1
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 50000KG/month
- Zinc hydroxide
-

- $0.00 / 1KG
-
2023-09-06
- CAS:20427-58-1
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 500000kg
|
| Product Name: | Zinc hydroxide | | Synonyms: | zincdihydroxide;zinchydroxide(zn(oh)2);ZINC HYDROXIDE;znmc hydroxide;Zinc dihydoxide;Zinc hydroxide≥ 99% (Assay);ZINC HYDROXIDE ISO 9001:2015 REACH | | CAS: | 20427-58-1 | | MF: | H2O2Zn | | MW: | 99.39 | | EINECS: | 243-814-3 | | Product Categories: | Inorganics | | Mol File: | 20427-58-1.mol | 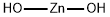 |
| | Zinc hydroxide Chemical Properties |
| Melting point | 125, decomposes [HAW93] | | density | 3.050 | | vapor pressure | 0Pa at 20℃ | | form | colorless orthorhombic crystals | | color | colorless orthorhombic crystals, crystalline | | PH | 8.88(1 mM solution);8.88(10 mM solution);8.88(100 mM solution) | | Water Solubility | slightly soluble H2O [HAW93] | | Solubility Product Constant (Ksp) | pKsp: 16.5 | | Stability: | Stable. | | LogP | -1.380 (est) | | CAS DataBase Reference | 20427-58-1(CAS DataBase Reference) | | EPA Substance Registry System | Zinc hydroxide (20427-58-1) |
| | Zinc hydroxide Usage And Synthesis |
| Description | Zinc hydroxide is a white solid that usually forms in solution. It is not very stable as a dried solid, therefore a standard of zinc hydroxide is not available commercially.
Zinc hydroxide Zn(OH)2 is an amphoteric chemical compound. Both bases and acids react with zinc hydroxide. It's an insoluble hydroxide that dissolves when exposed to a strong acid. It occurs naturally as the rare minerals Wulfingite, ashoverite and sweetite. Zinc hydroxide is used as a biological absorbant often. | | Chemical Properties | Colorless crystals.Decomposes
at 125C. Almost insoluble in water; forms
both zinc salts and zincates. | | Physical properties | Colorless orthorhombic crystals; density 3.053 g/cm3; decomposes at 125°C; slightly soluble in water. | | Uses | Intermediate, absorbent in surgical dressings,
rubber compounding. | | Preparation | The compound is prepared by adding a strong alkali to a solution of zinc sulfate or chloride: ZnSO4 + 2NaOH → Zn(OH)2 + Na2SO4. |
| | Zinc hydroxide Preparation Products And Raw materials |
|