|
|
| | (1S,2R)-1-(2-(HYDROXY-3,5-DI-TERT-BUTYL& Basic information |
| Product Name: | (1S,2R)-1-(2-(HYDROXY-3,5-DI-TERT-BUTYL& | | Synonyms: | (1S,2R)-1-(2-(HYDROXY-3,5-DI-TERT-BUTYL&;(1S,2R)-1-[(3,5-Di-tert-butyl-2-hydroxybenzylidene)amino]-2-indanol 97%;(1S,2R)-1-((3,5-Di-tert-butyl-2-hydroxybenzylidene)amino)-2,3-dihydro-1H-inden-2-ol;1H-Inden-2-ol, 1-[[[3,5-bis(1,1-dimethylethyl)-2-hydroxyphenyl]methylene]amino]-2,3-dihydro-, (1S,2R)-;(1S,2R)-1-[(3,5-Di-tert-butyl-2-hydroxybenzylidene)amino]-2-... | | CAS: | 212378-89-7 | | MF: | C24H31NO2 | | MW: | 365.51 | | EINECS: | | | Product Categories: | | | Mol File: | 212378-89-7.mol | 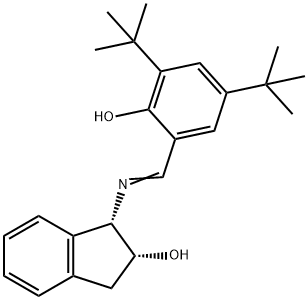 |
| | (1S,2R)-1-(2-(HYDROXY-3,5-DI-TERT-BUTYL& Chemical Properties |
| | (1S,2R)-1-(2-(HYDROXY-3,5-DI-TERT-BUTYL& Usage And Synthesis |
| Uses | (1S,2R)-1-[(3,5-Di-tert-butyl-2-hydroxybenzylidene)amino]-2-indanol can be used as a ligand to prepare Schiff-base chromium(III) complex, which is used as a catalyst in the synthesis of:
- Poly(cyclohexene carbonate) by coupling of CO2 and cyclohexene oxide.
- β-hydroxyenol ethers by asymmetric hetero-ene reaction between aryl aldehydes and 2-methoxypropene.
|
| | (1S,2R)-1-(2-(HYDROXY-3,5-DI-TERT-BUTYL& Preparation Products And Raw materials |
|