|
|
| | (-)-Indolmycin Basic information |
| Product Name: | (-)-Indolmycin | | Synonyms: | (S)-5-[(1R)-1-(1H-Indol-3-yl)ethyl]-2-(methylamino)-4(5H)-oxazolone;[5S,(-)]-5-[(R)-1-(1H-Indol-3-yl)ethyl]-2-methylamino-2-oxazolin-4-one;Pa-155-A;(-)-INDOLMYCIN;4(5H)-Oxazolone, 5-[(1R)-1-(1H-indol-3-yl)ethyl]-2-(methylamino)-, (5S)-;indolomycin | | CAS: | 21200-24-8 | | MF: | C14H15N3O2 | | MW: | 257.29 | | EINECS: | | | Product Categories: | | | Mol File: | 21200-24-8.mol | 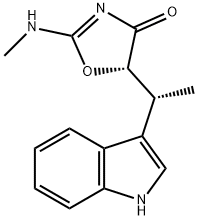 |
| | (-)-Indolmycin Chemical Properties |
| Melting point | 209-210° | | alpha | D25 -214° (c = 2 in methanol) | | Boiling point | 420.8±37.0 °C(Predicted) | | density | 1.36±0.1 g/cm3(Predicted) | | storage temp. | Store at -20°C, protect from light | | solubility | Soluble in ethanol;methanol;DMSO;dimethyl formamide | | form | powder | | pka | 16.65±0.30(Predicted) | | color | white |
| | (-)-Indolmycin Usage And Synthesis |
| Uses | Indolmycin is a highly selective antibiotic which acts as a tryptophan anti-metabolite. Recent research has shown that indolmycin is active against Mycobacteria and H. pylori, and can stimulate transcription in Escherichia coli. | | Uses | Indolmycin is an antibiotic that inhibits bacterial tryptophanyl-tRNA synthetase. | | Definition | ChEBI: A member of the class of 1,3-oxazoles that is 1,3-oxazol-4(5H)-one which is substituted at the 2 and 5-pro-S positions by methylamino and [(1R)-1-(1H-indol-3-yl)ethyl] gro
ps, respectively. | | Biological Activity | indolmycin is an antibiotic.tryptophanyl-trna synthetase (trprs) catalyzes activation of tryptophan through atp and transfer to trnatrp , leading to translation of the genetic code for tryptophan. | | in vitro | indolmycin was found to ba a bacteriostatic that showed good activity against methicillin-resistant s. aureus, methicillin-susceptible staphylococcus aureus, and vancomycin-intermediate s. aureus, including strains resistant to mupirocin or fusidic acid. spontaneous indolmycin-resistant mutants was observed at a lower frequency than those selected by mupirocin or fusidic acid and exhibited no cross-resistance with the comparative drugs. high-level resistance of indolmycin at its mic of 128 mg/l that was associated with an h43n mutation in tryptophanyl-trna synthetase, the target enzyme of indolmycin, led to loss of bacterial fitness. however, the locus responsible for low-level indolmycin resistance (indolmycin mics 8-32 mg/l) was not identified [1]. | | in vivo | animal study found that indolmycin could completely clear h. pylori in experimentally infected mongolian gerbils at a dose of 10 mg/kg. therefore, indolmycin could be regarded as a candidate for the treatment of h. pylori infection [2]. | | IC 50 | 30 μm for prokaryotic tryptophanyl-trna synthetase | | references | [1] hurdle jg, o'neill aj, chopra i. anti-staphylococcal activity of indolmycin, a potential topical agent for control of staphylococcal infections. j antimicrob chemother. 2004 aug;54(2):549-52. epub 2004 jul 8.
[2] vecchione jj, sello jk. a novel tryptophanyl-trna synthetase gene confers high-level resistance to indolmycin. antimicrob agents chemother 2009; 53: 3972-3980. |
| | (-)-Indolmycin Preparation Products And Raw materials |
|