| Company Name: |
Sigma-Aldrich
|
| Tel: |
021-61415566 800-8193336 |
| Email: |
orderCN@merckgroup.com |
| Products Intro: |
Product Name:4-Nitrophenyl 4-guanidinobenzoate hydrochloride
CAS:19135-17-2
Purity:protease inhibitor and substrate Package:500MG Remarks:N8010-500MG
|
| Company Name: |
Energy Chemical
|
| Tel: |
021-58432009 400-005-6266 |
| Email: |
marketing@energy-chemical.com |
| Products Intro: |
Product Name:4-Nitrophenyl 4-guanidinobenzoate hydrochloride protease inhibitor and substrate
CAS:19135-17-2
Purity:protease inhibitor and substrate Package:1g;500mg Remarks:NULL
|
|
| Product Name: | NPGB | | Synonyms: | 4-GUANIDINOBENZOIC ACID 4-NITROPHENYL-ESTER HYDROCHLORIDE;4-NITROPHENYL 4-GUANIDINOBENZOATE HYDROCHLORIDE;NPGB;PNPGB;P-NITROPHENYL P'-GUANIDINOBENZOATE HYDROCHLORIDE;P-NITROPHENYL-P-GUANIDINOBENZOATE HYDROCHLORIDE;P'-GUANIDINOBENZOIC ACID P-NITROPHENYL ESTER HYDROCHLORIDE;Nsc163801 | | CAS: | 19135-17-2 | | MF: | C14H13ClN4O4 | | MW: | 336.73 | | EINECS: | 242-831-3 | | Product Categories: | | | Mol File: | 19135-17-2.mol | 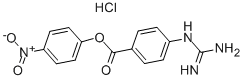 |
| Melting point | 241-243 °C | | storage temp. | -20°C | | form | Solid | | color | White to off-white | | BRN | 8024554 |
| Hazard Codes | Xi | | Risk Statements | 36 | | Safety Statements | 26 | | WGK Germany | 3 | | F | 8-10 |
| Uses | 4-Nitrophenyl 4-guanidinobenzoate hydrochloride has been used:
- as a substrate for trypsin for active site titration experiments
- for pre-treating of mosquito eggs in the interplasmid transposition assay
- as a component of isotonic buffer to moisten filter paper for mosquito embryo collection
| | General Description | 4-Nitrophenyl 4-guanidinobenzoate hydrochloride (pNPGB) is a trypsin substrate. It is more water-soluble than 4-methylumbelliferyl 4-guanidinobenzoate and is preferred in spectrophotometric assay. | | Biochem/physiol Actions | 4-Nitrophenyl 4-guanidinobenzoate hydrochloride (pNPGB) inhibits phenoloxidase enzyme and delays embryo chorionic membrane hardening. | | References | [1] BHUMI PATEL . L. donovani XPRT: Molecular characterization and evaluation of inhibitors[J]. Biochimica et biophysica acta. Proteins and proteomics, 2018, 1866 3: Pages 426-441. DOI:10.1016/j.bbapap.2017.12.002.
[2] FRANK S. STEVEN Margaret M G. Studies on the Molecular Mechanism of Mersalyl and 4-Aminophenylmercuric Acetate Re-activation of Trypsin-Thiol Complexes[J]. The FEBS journal, 1980, 109 2: 567-573. DOI:10.1111/j.1432-1033.1980.tb04829.x.
[3] YING-YING HE . Isolation, expression and characterization of a novel dual serine protease inhibitor, OH-TCI, from king cobra venom[J]. Peptides, 2008, 29 10: Pages 1692-1699. DOI:10.1016/j.peptides.2008.05.025.
[4] J.A. SCHIFFERLI G. S. A simple two-step procedure for the preparation of the first component of human complement (C1) in its native form[J]. Journal of immunological methods, 1985, 76 2: Pages 283-288. DOI:10.1016/0022-1759(85)90305-9.
[5] J. M. KAMINSKI. Synthesis and inhibition of human acrosin and trypsin and acute toxicity of aryl 4-guanidinobenzoates[J]. Journal of Medicinal Chemistry, 1986, 29 4: 514-519. DOI:10.1021/jm00154a015. |
| | NPGB Preparation Products And Raw materials |
|