Fenipentol manufacturers
- Fenipentol
-
- $0.00 / 1KG
-
2024-09-12
- CAS:583-03-9
- Min. Order: 1KG
- Purity: 99%+
- Supply Ability: 100000kgs per Month
- Fenipentol
-

- $0.00 / 1g
-
2019-12-31
- CAS:583-03-9
- Min. Order: 1g
- Purity: 95%min
- Supply Ability: 20kg/month
|
| | Fenipentol Basic information | | Aroma |
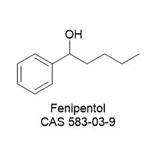
| Product Name: | Fenipentol | | Synonyms: | 1-PHENYLHYDROXYPENTANE;2-(4-Hydroxybenzoyloxy)-N,N,N-trimethylethanaminium;p-Hydroxybenzoylcholine;BenzeneMethanol, a-butyl-;D52;N8L;PrLZ;1-Hydroxy-1-phenylpentane | | CAS: | 583-03-9 | | MF: | C11H16O | | MW: | 164.25 | | EINECS: | 209-493-9 | | Product Categories: | 583-03-9 | | Mol File: | 583-03-9.mol | 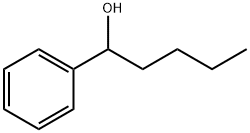 |
| | Fenipentol Chemical Properties |
| Safety Statements | 24/25 | | RTECS | DN8780000 | | TSCA | Yes | | HS Code | 2906.29.6000 |
| Provider | Language |
|
ALFA
| English |
| | Fenipentol Usage And Synthesis |
| Aroma | Mild floral odor, slightly green, woody-
barklike. One manufacturer classifies Fenipentol as
"Orris-like".
Useful as a fixative in Lilac or as a modifierfixative for compositions where Linalool is
present. | | Physical properties | White solid lumps or crystalline powder.
M.P. 69°C. B.P. 285°C.
Insoluble in water, soluble in alcohol and
oils. | | Originator | Pancoral,Eisai,Japan,1973 | | Uses | Biliary dyskinesia;Choleretic | | Definition | ChEBI: Fenipentol is a member of benzenes. | | Production Methods | Fenipentol can be produced by reduction of Valerophenone with
Sodium. | | Manufacturing Process | The 1-phenylpentanol-(1) may be prepared in any convenient manner.
Benzaldehyde may be reacted with n-butyl-magnesium bromide, and after
purification 1-phenyl-pentanol-(1) is obtained in the form of a colorless oil at
room temperature. | | Therapeutic Function | Choleretic | | Synthesis Reference(s) | Tetrahedron Letters, 25, p. 5187, 1984 DOI: 10.1016/S0040-4039(01)81559-8 | | Safety Profile | Poison by
intraperitoneal route. Moderately toxic by
ingestion and subcutaneous routes. An
experimental teratogen. Other experimental
reproductive effects. Stimulates the
production of bile by the liver. When heated
to decomposition it emits acrid smoke and
irritating fumes. |
| | Fenipentol Preparation Products And Raw materials |
|