BENZIMIDAVIR manufacturers
- Maribavir
-

- $0.00 / 1kg
-
2025-02-14
- CAS:176161-24-3
- Min. Order: 1kg
- Purity: 98
- Supply Ability: 1000
- Maribavir
-

- $36.00 / 1mg
-
2024-11-19
- CAS:176161-24-3
- Min. Order:
- Purity: 99.85%
- Supply Ability: 10g
|
| | BENZIMIDAVIR Basic information |
| Product Name: | BENZIMIDAVIR | | Synonyms: | 5,6-Dichloro-N-(1-methylethyl)-1-beta-L-ribofuranosyl-1H-benzimidazol-2-amine;BW 1263W94;GW 257406X;(2S,3S,4R,5S)-2-(5,6-Dichloro-2-(isopropylamino)-1H-benzo[d]imidazol-1-yl)-5-(hydroxymethyl)tetrahydrofuran-3,4-diol;(2S,3S,4R,5S)-2-(5,6-Dichloro-2-(isopropylamino)-1H-benzo[d]-imidazol-1-yl)-5-(hydroxymethyl)t;5,6-DICHLORO-2-(ISOPROPYLAMINO)-1-(BETA-L-RIBOFURANOSYL)-1H-BENZIMIDAZOLE;BENZIMIDAVIR;BENZIMIDAVIR, 5,6-DICHLORO-2-(ISOPROPYLAMINO)-1-(β-L-RIBOFURANOSYL)-LH-BENZIMIDAZOLE | | CAS: | 176161-24-3 | | MF: | C15H19Cl2N3O4 | | MW: | 376.24 | | EINECS: | | | Product Categories: | Inhibitors | | Mol File: | 176161-24-3.mol | 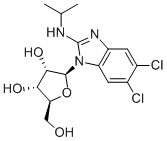 |
| | BENZIMIDAVIR Chemical Properties |
| Boiling point | 611.0±65.0 °C(Predicted) | | density | 1.67±0.1 g/cm3 (20 ºC 760 Torr) | | storage temp. | 2-8°C(protect from light) | | solubility | Soluble in DMSO | | form | Powder | | pka | 13.20±0.70(Predicted) | | color | White to off-white |
| | BENZIMIDAVIR Usage And Synthesis |
| Description | Maribavir is an orally bioavailable benzimidazole L-riboside antiviral, with a spectrum of activity essentially limited to human cytomegalo�virus (CMV) and Epstein–Barr virus (EBV). It is an inhibitor of the CMV UL97 kinase. As of 2008, maribavir was in phase III clinical trials for prevention of CMV infection in transplant recipients at risk. Earlier phase I and II trials of maribavir showed anti-CMV activity with an acceptable adverse effect profile. Investigational drug code names include 1263W94, BW1263-W94, GW257406X, and VP41263, reflecting changes in ownership during drug development. As of early 2009, the drug was being developed under ‘‘fast track’’ status granted by the US Food and Drug Administration, but the apparent failure of maribar prophylaxis to prevent CMV infection in stem cell transplant recipients in the definitive phase III clinical trial resulted in the sponsor halting ongoing clinical development. | | Uses | Treatment of cytomegalovirus infections
(antiviral). | | Mechanism of action | Maribavir inhibits the CMV UL97 kinase, an
enzyme which is required for the normal replication of the virus . In the absence of functioning UL97 kinase,
viral replication is severely impaired in vitro, with an abnormal cell
culture cytopathic effect characterized by the nuclear aggregation of
excess amorphous viral proteins, mainly the tegument protein pp65. Impaired UL97 function appears to cause a
defect in viral encapsidation and/or egress of viral
particles from the nucleus. In addition, viral DNA
synthesis may also be reduced.
CMV replication is not completely shut off in the absence of the UL97
kinase; the widely varying maribavir IC50s under different assay
conditions suggest that host cells can variably substitute for the normal
function of UL97, a factor that may affect the therapeutic potency of
maribavir in vivo. | | Drug interactions | Since CYP3A4 appears to be the major maribavir-metabolizing
enzyme, there are potential drug interactions with CYP3A4 inhibitors,
such as azole antifungals, macrolide antibiotics, and HIV protease
inhibitors, or CYP3A4 inducers, such as rifampicin or efavirenz. There
are insufficient data to assess the clinical significance of this
interaction. Phase I clinical trials examined the pharmacokinetics of
maribavir in HIV-infected subjects, many of whom were concomitantly
using antifungals and protease inhibitors. Overall, the pharmacokinetic data were not significantly
different from those of healthy individuals not on these drugs. In
healthy adults, oral administration of ketoconazole, a potent CYP3A4
inhibitor, resulted in a 35% decrease in the clearance of maribavir, which is not expected to have adverse
consequences because of the low observed toxicity of maribavir.
Further studies are required to assess potential impairment of antiviral
activity by CYP3A4 inducers.
Maribavir may have some inhibitory effect on cytochrome P450
isozymes, CYP2C19 and CYP2D6, as assessed after administration of
multiple concurrently administered drug probes, including omeprazole
(2C19) and dextromethorphan (2D6). |
| | BENZIMIDAVIR Preparation Products And Raw materials |
|