|
|
| | 2-(Diphenylphosphino)benzoic acid Basic information |
| | 2-(Diphenylphosphino)benzoic acid Chemical Properties |
| | 2-(Diphenylphosphino)benzoic acid Usage And Synthesis |
| Chemical Properties | light yellow powder | | Uses | The ligand (R ,R )‐1,2‐Bis(aminocarbonylphenyl‐2′‐diphenylphosphino)cyclohexane[138517-61-0] can be prepared by the coupling of (1R ,2R )‐(-)‐1,2‐diaminocyclohexane [20439-47-8] with 2‐(diphenylphosphino)benzoic acid [17261-28-8], using reagents such as DCC.
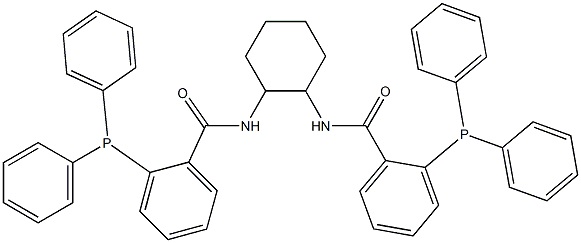
An alternative procedure has been developed where by (1R ,2R )‐(+)‐1,2‐diaminocyclohexane L‐tartrate salt [39961-95-0] is coupled to a mixed anhydride of 2‐(diphenylphosphino)benzoic acid and diphenylchlorophosphate.The procedure is reproduced below 2‐(Diphenylphosphino)benzoic acid (20 g, 65.3 mmol, 2 equiv) is suspended in dichloromethane (150 mL) and cooled in an ice‐water bath to 0°C (internal temperature). Triethylamine (10.1 mL, 71.8 mmol, 2.2 equiv) is added dropwise and a clear solution is obtained. This process is exothermic and a rise in temperature to 5°C is observed. The solution is re‐cooled to 0°C and diphenylchlorophosphate (13.4 mL, 64.7 mmol, 1.98 equiv) is added slowly, maintaining the internal temperature between 0–5°C. The yellow solution is stirred for 1 h at 0°C. (1R, 2R )‐(+)‐1,2‐Diaminocyclohexane‐L‐tartrate salt (8.63 g, 32.65 mmol, 1 equiv) is suspended in water (50 mL, 5.8 vol) and potassium carbonate (15 g, 107.8 mmol, 3.3 equiv) is added. This process is exothermic and a clear solution is obtained after approximately 10 min. After 30 min, the clear aqueous solution of diamine is added to the mixed anhydride solution at 0°C, and the resulting yellow two‐phase mixture is stirred for 2 h at 0°C, then allowed to warm to room temperature. After 14 h, the mixture is poured into a separating funnel and 200 mL of dichloromethane and 100 mL of water are added. The organic phase is separated, washed with 2 N HCl (100 mL) and saturated aqueous NaHCO3 solution (100 mL), then dried over magnesium sulfate. The dried organic phase is filtered through a silica pad and the pad is washed with dichloromethane (50 mL). The combined filtrates are evaporated to dryness under reduced pressure, producing a yellow foam (22.3 g, 99% crude). The foam is crystallized from boiling acetonitrile (390 mL, 17.5 vol) to afford a white crystalline solid. The solid is dried under vacuum to provide the phosphine ligand (15 g, 67%). | | Uses | 2-(Diphenylphosphino)benzoic Acid is used in small molecule control of protein function through staudinger reduction. | | reaction suitability | reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling |
| | 2-(Diphenylphosphino)benzoic acid Preparation Products And Raw materials |
|