|
|
| | 1,4-Bis(vinyloxy)-butane Basic information |
| Product Name: | 1,4-Bis(vinyloxy)-butane | | Synonyms: | 1,4-DIVINYLOXYBUTANE;1,4-BUTYLENE GLYCOL DIVINYL ETHER;1,4-BUTYLDIOL VINYL ETHER;1,4-BUTANEDIOL DIVINYL ETHER;1,4-BIS-(VINYLOXY)-BUTANE;1,4-bis(ethenyloxy)-butan;1,4-Bis(ethenyloxy)butane;1,4-divinyloxybutane(1,4-butanedioldivinylether) | | CAS: | 3891-33-6 | | MF: | C8H14O2 | | MW: | 142.2 | | EINECS: | 223-437-0 | | Product Categories: | Monomers;Polymer Science;Vinyl Ethers | | Mol File: | 3891-33-6.mol | 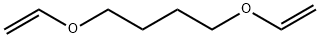 |
| | 1,4-Bis(vinyloxy)-butane Chemical Properties |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36 | | WGK Germany | 3 |
| | 1,4-Bis(vinyloxy)-butane Usage And Synthesis |
| Uses |
1,4-Bis(vinyloxy)-butane, which also serves as an intermediate in manufacturing higher perfume chemicals, has found some use in perfume compositions, mainly for its diffusive power and “lift” in soap fragrances, detergent perfumes, etc. It seems most conceivable that certain Vinyl ethers are the intermediate steps made by Mother Nature in the manufacture (biosynthesis) of some very interesting and important odorous chemicals, including “Rose oxide”. Many Pyran derivatives are made by means of Vinylether synthesis.
| | Production Methods | 1,4-Bis(vinyloxy)-butane was produced from Tetramethylene glycol and Acetylene, using pressure and a catalyst. | | Flammability and Explosibility | Flammable | | Solubility in water |
Insoluble in water, soluble in alcohol, miscible with oils.
|
| | 1,4-Bis(vinyloxy)-butane Preparation Products And Raw materials |
|