| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
010-82848833 400-666-7788 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:PrednisolaMate
CAS:5626-34-6
Package:100Mg,1g
|
| Company Name: |
ZHIWE CHEMTECH CO LTD
|
| Tel: |
021-20221225 13917446399 |
| Email: |
sales@zhiwe.net |
| Products Intro: |
CAS:5626-34-6
|
| Company Name: |
Energy Chemical
|
| Tel: |
021-58432009 400-005-6266 |
| Email: |
marketing@energy-chemical.com |
| Products Intro: |
Product Name:Prednisolamate
CAS:5626-34-6
Purity:NULL Package:100mg;1g Remarks:NULL
|
| Company Name: |
Shaanxi Dideu Newmaterial Co., Ltd.
|
| Tel: |
029-87576359 15353716720 |
| Email: |
1039@dideu.com |
| Products Intro: |
Product Name:prednisolamate
CAS:5626-34-6
Purity:90% Package:25kg
|
|
| | prednisolamate Basic information |
| Product Name: | prednisolamate | | Synonyms: | Prednisolone 21-Diethylaminoacetate;N,N-Diethylglycine 11β,17-dihydroxy-3,20-dioxopregna-1,4-dien-21-yl ester;prednisolamate;ILZSJEITWDWIRX-FOMYWIRZSA-N;Glycine, N,N-diethyl-, (11β)-11,17-dihydroxy-3,20-dioxopregna-1,4-dien-21-yl ester | | CAS: | 5626-34-6 | | MF: | C27H39NO6 | | MW: | 473.6 | | EINECS: | 227-064-4 | | Product Categories: | | | Mol File: | 5626-34-6.mol | 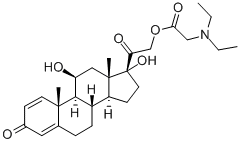 |
| | prednisolamate Chemical Properties |
| Melting point | 175-177° | | storage temp. | -20°C Freezer | | solubility | Dioxane (Slightly), DMSO (Slightly) | | form | Solid | | color | Pale Beige to Light Brown |
| | prednisolamate Usage And Synthesis |
| Originator | Prednisolamate ,Pfizer and Co. | | Uses | Prednisolamate is a derivative of Prednisone (P703780), a glucocorticoid hormone that is used to treat patients with asthma, and to improve renal function in human immunodeficiency virus-associated nephropathy. | | Definition | ChEBI: Prednisolamate is a corticosteroid hormone. | | Manufacturing Process | To a solution of 30 g prednisolone and 10.2 ml pyridine in 99 ml dimethylformamide, cooled to 0°C in an ice bath and protected from atmospheric moisture, was added dropwise with stirring 9.6 ml chloroacetyl chloride. Stirring was continued for 1 hour at 0°C, then stopped and the reaction allowed to come to room temperature and stand overnight. The following morning the reaction mixture was poured with vigorous stirring into a vessel containing 360 ml 1 N sulfuric acid, and the product washed with fresh portions of water to neutral wash was obtained. The product was thoroughly dried in a vacuum desiccator and recrystallized from isopropyl alcohol, using 200 ml solvent. The yield was better than 80%, and the compound had MP: 240.6°-242.8°C and [α]D = +114.6° (Dioxane).
Two grams of this prednisolone chloracetate and 40 ml of colorless, freshly distilled diethylamine were refluxed, with stirring, under nitrogen for one hour. The excess diethylamine was removed in vacuum at room temperature. The residue was taken up in 100 ml CHCl3, and a small amount of water; the CHCl3, was washed with one 50 ml portion of 5% aqueous sodium bicarbonate and two 50 ml portions of water, and dried over sodium sulfate. The chloroform was then concentrated to dryness in vacuum. The residue was recrystallized from acetone-hexane yielded prednisolone 21-diethylaminoacetate 1.67 g; MP: 175.0°-197.2°C. This product (1 g) was suspended in a mixture of 15 ml acetone and 1.5 ml chloroform. The suspension was cooled to 0°C in an ice bath, and with vigorous stirring the ethereal HCl solution was slowly added until the resulting mixture gave an acid reaction to the congo red paper. The product was removed by filtration and recrystallized from ethanol. The yield was approximately 80%; MP: 239.4°-239.8°C; [α]D = +120.7° (water). The product was the hydrochloride salt of the prednisolone-N,Ndiethylaminoacetate, very active therapeutically and excellently watersoluble. | | Therapeutic Function | Glucocorticoid |
| | prednisolamate Preparation Products And Raw materials |
|