(2-BIPHENYL)DI-TERT-BUTYLPHOSPHINE GOLD& manufacturers
|
| | (2-BIPHENYL)DI-TERT-BUTYLPHOSPHINE GOLD& Basic information | | Reaction |
| Product Name: | (2-BIPHENYL)DI-TERT-BUTYLPHOSPHINE GOLD& | | Synonyms: | (2-BIPHENYL)DI-TERT-BUTYLPHOSPHINE GOLD&;Chloro[2-(di-t-butylphosphino)biphenyl]gold(I),99%;1,1''-BIPHENYL]-2-YLBIS(T-BUTYLPHOSPHINE]CHLOROGOLD;(2-Biphenyl)di-tert-butylphosphine gold(I) chloride, 2-(Di-tert-butylphosphino)biphenyl gold(I) chloride, [[1,1μ-Biphenyl]-2-ylbis(t-butylphosphine]chlorogold;Chloro[(1,1μ-biphenyl-2-yl)di-tert-butylphosphine]gold(I);Chloro[2-(di-t-butylphosphino)biphenyl]gold(I), 99%;(2-Biphenyl)di-tert-butylphosphine gold(I) chloride;2-(Di-tert-butylphosphino)biphenyl gold(I) chloride | | CAS: | 854045-93-5 | | MF: | C20H28AuClP | | MW: | 531.83 | | EINECS: | | | Product Categories: | Au;organometallic complex | | Mol File: | 854045-93-5.mol | 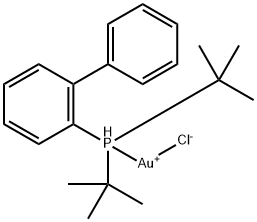 |
| | (2-BIPHENYL)DI-TERT-BUTYLPHOSPHINE GOLD& Chemical Properties |
| Melting point | 237-240 °C | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | form | Powder | | color | white |
| | (2-BIPHENYL)DI-TERT-BUTYLPHOSPHINE GOLD& Usage And Synthesis |
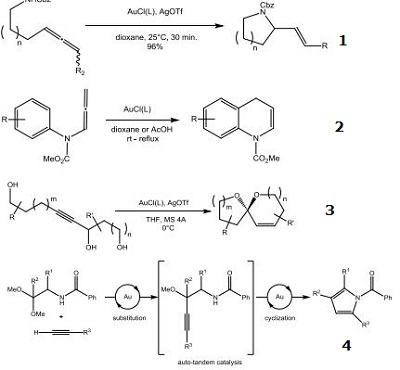
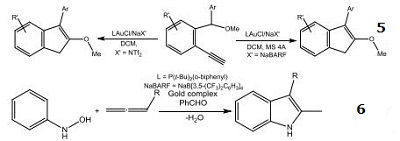
| Reaction |
- Highly active gold catalyst for the intramolecular exohydrofunctionalization of allenes.
- Catalyst used for the hydroarylation of allenes.
- Catalyst used for the intramolecular cyclization of monopropargyl triols.
- Synthesis of pyrroles via a gold-catalyzed cascade reaction.
- Gold-catalyzed carboalkoxylations of 2-ethynylbenzyl ethers.
- Gold-catalyzed annulations of allenes with N-hydroxy anilines.
| | Uses | Gold Catalysts — 21st Century ′Gold Rush′ |
| | (2-BIPHENYL)DI-TERT-BUTYLPHOSPHINE GOLD& Preparation Products And Raw materials |
|