|
|
| | amitriptyline N-oxide Basic information |
| Product Name: | amitriptyline N-oxide | | Synonyms: | amitriptyline N-oxide;Amitriptylinoxide (inn);D07449;ZPMKQFOGINQDAM-UHFFFAOYSA-N;Nortriptyline EP Impurity J;1-?Propanamine, 3-?(10,?11-?dihydro-?5H-?dibenzo[a,?d]?cyclohepten-?5-?ylidene)?-?N,?N-?dimethyl-?, N-?oxide;Amitriptyline N-Oxide (Nortriptyline EP Impurity J);Nortriptyline Impurity 10 (Nortriptyline EP Impurity J) | | CAS: | 4317-14-0 | | MF: | C20H23NO | | MW: | 293.4 | | EINECS: | | | Product Categories: | | | Mol File: | 4317-14-0.mol | 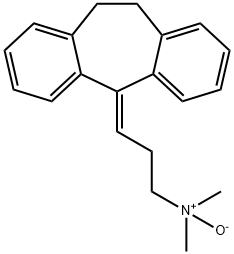 |
| | amitriptyline N-oxide Chemical Properties |
| Melting point | 228-230°; mp 102-103° | | Boiling point | 435.25°C (rough estimate) | | density | 1.0552 (rough estimate) | | refractive index | 1.6000 (estimate) | | storage temp. | Amber Vial, -86°C Freezer, Under inert atmosphere | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Solid | | color | White to Off-White | | Stability: | Light and Temperature Sensitive |
| Toxicity | LD50 in mice, guinea pigs, rabbits, dogs (mg/kg): between 330-460 orally; in mice, rats: 87, 25 i.v.; 320, 110 i.p. (Friehe, Fontaine). |
| | amitriptyline N-oxide Usage And Synthesis |
| Originator | Equilibrin,Nattermann,W. Germany,1980 | | Uses | Amitriptyline N-oxide is used in biological studies to metabolize drugs by fungi colonizing cadavers using LC-ESI-MS/MS and GC-MS. This compound is also used in the development of antidepressant drugs. | | Definition | ChEBI: Amitriptylinoxide is an organic tricyclic compound. | | Manufacturing Process | 31.3 g (0.1 mol) of 3-(3'-dimethylaminopropylidene)dibenzo[a,d]cyclohepta-
1,4-diene hydrochloride are dissolved in water, and the free base is liberated
by means of a 28% aqueous solution of sodium hydroxide. The free base is
sucked off, washed with water, and dissolved in 100 ml of methanol. To the
solution are added 31 ml of 30% hydrogen peroxide. After 7 days, the
reaction mixture is diluted with 200 ml of water, and the major part of the
methanol is evaporated in vacuum. The precipitated N-oxide crystals are
filtered off, washed with water, and dried, yielding 27 g of the dihydrate of 3-
(3'-dimethylaminopropylidene)dibenzo[a,d]cyclohepta-1,4-diene N-oxide with
melting point of 102° to 103°C. In dehydrated state the melting point is
228°C to 230°C.
By dissolving the N-oxide in acetone, and bubbling dry hydrogen chlorine gas
through the solution until slightly acid reaction, the hydrochloride of the Noxide
is precipitated as a white crystalline substance with melting point of
172°C to 173.6°C.
The starting material can be prepared in known manner from
dibenzo[a,d]cyclohepta-1,4-diene-5-one by a Grignard reaction with 3-
dimethylaminopropyl magnesium chloride, hydrolysis and dehydration of the
resulting carbinol. | | Therapeutic Function | Antidepressant | | Safety Profile | Poison by ingestion,intraperitoneal, and intravenous routes. An experimentalteratogen. Other experimental reproductive effects. Whenheated to decomposition it emits toxic fumes of NOx. |
| | amitriptyline N-oxide Preparation Products And Raw materials |
|