2-IMINOBIOTIN HYDROBROMIDE manufacturers
|
| | 2-IMINOBIOTIN HYDROBROMIDE Basic information |
| Product Name: | 2-IMINOBIOTIN HYDROBROMIDE | | Synonyms: | GUANIDINOBIOTIN;HEXAHYDRO-2-IMINO-1H-THIENO[3,4-D]IMIDAZOLE-4-PENTANOIC ACID;2-IMINOBIOTIN HYDROBROMIDE;2-Iminobiotine;2-IMINOBIOTIN 98+%;Guanidinobiotin, Hexahydro-2-imino-1H-thieno[3,4-d]imidazole-4-pentanoic acid;(3aR,6S,6aS)-2-Amino-3a,4,6,6a-tetrahydro-1H-thieno[3,4-d]imidazole-6-pentanoic acid;Iminobiotin | | CAS: | 13395-35-2 | | MF: | C10H17N3O2S | | MW: | 243.33 | | EINECS: | | | Product Categories: | | | Mol File: | 13395-35-2.mol | 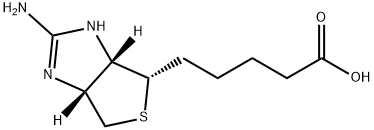 |
| | 2-IMINOBIOTIN HYDROBROMIDE Chemical Properties |
| Boiling point | 477.3±55.0 °C(Predicted) | | density | 1.60 | | storage temp. | 2-8°C | | solubility | 1 M HCl: 50 mg/mL, clear, colorless | | form | White to off-white powder. | | pka | 4.73±0.10(Predicted) | | color | White to off-white | | BRN | 18952 |
| | 2-IMINOBIOTIN HYDROBROMIDE Usage And Synthesis |
| Uses | 2-Iminobiotin has been used as a competitive inhibitor to bind to biotic binding sites on streptavidin in order to restore fluorescence signal. | | General Description | 2-Iminobiotin is a cyclic guanidino analog of biotin that has high affinity for avidin at high pH (>9) and low affinity at low pH (<6). The proteins that are iminobiotinylated are selectively contained within the avidin column at pH 9 to 11 and may be eluted at pH 4 or by the addition of biotin. This technique may be very useful in the study of cell surface proteins by labeling and selectively recovering the components that are periodate-sensitive from intact human erythrocyte surface. 2-Iminobiotin reversibly inhibits NOS (nitric oxide synthases) via its guanidino group. It inhibits NO biosynthesis and may be important to understand the binding-site interactions for this important class of enzymes. NOS oxidizes the guanidino-nitrogen of L-arginine to produce nitric oxide and L-citrulline. |
| | 2-IMINOBIOTIN HYDROBROMIDE Preparation Products And Raw materials |
|