| | Potassium superoxide Basic information |
| Product Name: | Potassium superoxide | | Synonyms: | PotassiuM Superoxide, Uncatalyzed Granules;PotassiuM Superoxide, Uncatalyzed Granules, 3/8 to 1/4 in.;Potassium dioxide chunks, 5-10 mm;burntpotash;cacinedpotash;K(O2);Potassium oxide super;Potassium superoxide ko2 | | CAS: | 12030-88-5 | | MF: | KO2* | | MW: | 71.1 | | EINECS: | 234-746-5 | | Product Categories: | metal oxide | | Mol File: | 12030-88-5.mol | 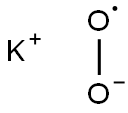 |
| | Potassium superoxide Chemical Properties |
| Melting point | 400 °C | | density | 2,14 g/cm3 | | storage temp. | Store at +15°C to +25°C. | | solubility | Soluble in ethanol and ether. | | form | powder | | color | Yellow-green | | Specific Gravity | 2.14 | | PH | >7 (H2O, 20℃) | | Water Solubility | reacts | | Sensitive | Air & Moisture Sensitive | | Stability: | Stable, but reacts violently with water. Incompatible with moisture, alcohols, strong reducing agents, strong acids, finely powdered metals. | | CAS DataBase Reference | 12030-88-5(CAS DataBase Reference) | | NIST Chemistry Reference | Potassium superoxide(12030-88-5) | | EPA Substance Registry System | Potassium superoxide (K(O2)) (12030-88-5) |
| Hazard Codes | O,C | | Risk Statements | 8-14-34-35 | | Safety Statements | 17-27-36/37/39-8-45-26 | | RIDADR | UN 2466 5.1/PG 1 | | WGK Germany | 3 | | RTECS | TT6053000 | | TSCA | Yes | | HazardClass | 5.1 | | PackingGroup | I | | HS Code | 28259080 |
| | Potassium superoxide Usage And Synthesis |
| Chemical Properties | light yellow powder or chunks | | Uses | Potassium oxide is used as a carbon dioxide scrubber, water dehumidifier and oxygen generator. It finds application in rebreathers for fighting with fire and mine rescue work. It is also used in spacecraft, submarines and spacesuit life support systems. | | Uses | Reagent and intermediate.
One use of potassium superoxide,KO2, is for generating oxygen. It has the ability to absorb carbon dioxide, while giving out oxygen at the same time:
4KO2(s)+ 2CO2(g)--->2K2CO3(s)+ 3O2(g)
This property has been made use of in breathing equipment,e.g.for mountaineers, in submarines and in spacecraft. | | General Description | A yellowish to white solid. Melting point 948°F. Mixtures with combustible material readily ignite by friction, heat, or contact with moisture. Prolonged exposure to fire or heat may cause vigorous decomposition of the material and rupturing of the container. | | Air & Water Reactions | Reacts explosively with water [Mellor 2, Supp. 3: 1631. 1963]. | | Reactivity Profile | Potassium superoxide is a powerful oxidizer. Forms on the surface of potassium metal, solid or molten, that is exposed to the air. Attempts to extinguish burning potassium with powdered graphite has resulted in violent explosions [Chem. Abstr. 63:424. 1965]. Highly oxidized potassium metal was dropped into a dish of ethyl alcohol, an immediate explosion shattered the dish. Potassium superoxide was considered the cause of the reaction [Health and Safety Inf. 251. 1967]. Potassium superoxide should not be added to pure organic materials (hydrocarbons), as ignition and violent explosion may occur. Oxidation of arsenic, antimony, copper, potassium, tin, or zinc proceeds with incandescence, [Mellor, 1941, Vol. 2, 493]. Interaction between the superoxide and diselenium dichloride is violent, [Mellor, 1947, Vol. 10, 897]. | | Hazard | Corrosive to tissue. | | Health Hazard | TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Fire may produce irritating and/or toxic gases. Toxic fumes or dust may accumulate in confined areas (basement, tanks, hopper/tank cars, etc.). Runoff from fire control or dilution water may cause pollution. | | Fire Hazard | May explode from friction, heat or contamination. These substances will accelerate burning when involved in a fire. May ignite combustibles (wood, paper, oil, clothing, etc.). Some will react explosively with hydrocarbons (fuels). Containers may explode when heated. Runoff may create fire or explosion hazard. | | Safety Profile | Explosive reaction
when heated with carbon, 2-aminophenol +
tetrahydrofuran (at 65°C). Forms a friction-
sensitive explosive mixture with
hydrocarbons. Violent reaction with
lselenium dichloride, ethanol, potassium-
sodium alloy. May ignite on contact with
organic compounds. Incandescent reaction
with metals (e.g., arsenic, antimony, copper,
potassium, tin, and zinc). When heated to
decomposition it emits toxic fumes of K2O.
See also PEROXIDES. |
| | Potassium superoxide Preparation Products And Raw materials |
|