|
|
| | Moxifloxacin Impurity Basic information |
| Product Name: | Moxifloxacin Impurity | | Synonyms: | 1-cyclopropyl-7-ethoxy-6-fluoro-8-
methoxy-4-oxo-1,4-dihydroquinoline-
3-carboxylic acid;1-cyclopropyl-7-ethoxy-6;Moxifloxacin-18;Moxifloxacin impurity 27/1-cyclopropyl-7-ethoxy-6-fluoro-8-methoxy-4-oxo-1,4-dihydroquinoline-3-carboxylic acid;Moxifloxacin Impurity 36;3-Quinolinecarboxylic acid, 1-cyclopropyl-7-ethoxy-6-fluoro-1,4-dihydro-8-methoxy-4-oxo-;Moxifloxacin Impurity 121;Azilsartan medoxomil impurity273 | | CAS: | 1797982-51-4 | | MF: | C16H16FNO5 | | MW: | 321.3 | | EINECS: | | | Product Categories: | | | Mol File: | 1797982-51-4.mol | 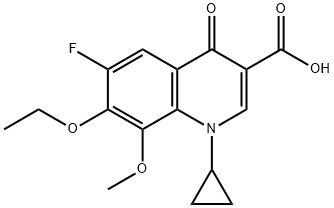 |
| | Moxifloxacin Impurity Chemical Properties |
| Boiling point | 505.2±50.0 °C(Predicted) | | density | 1.441±0.06 g/cm3(Predicted) | | pka | 6.40±0.50(Predicted) |
| | Moxifloxacin Impurity Usage And Synthesis |
| Uses | 1-Cyclopropyl-6-fluoro-1,4-dihydro-7-ethoxy-8-methoxy-4-oxo-3-quinolinecarboxylic Acid is used in the synthesis of new tetracyclic quinolone antibacterials. |
| | Moxifloxacin Impurity Preparation Products And Raw materials |
|