|
|
| | 4,6-DIACETYLRESORCINOL Basic information |
| Product Name: | 4,6-DIACETYLRESORCINOL | | Synonyms: | Ethanone, 1,1'-(4,6-dihydroxy-1,3-phenylene)bis-;4,6-DIACETYLRESORCINOL;AKOS 236-35;1,1'-(4,6-DIHYDROXY-1,3-PHENYLENE)BISETHANONE;1,1'-(4,6-dihydroxy-1,3-phenylene)bisethan-1-one;1-(5-ACETYL-2,4-DIHYDROXYPHENYL)ETHAN-1-ONE;RESODIACETOPHENONE;1,1'-(4,6-Dihydroxy-1,3-phenylene)bisethanone 99% | | CAS: | 2161-85-5 | | MF: | C10H10O4 | | MW: | 194.18 | | EINECS: | 218-482-8 | | Product Categories: | Aromatic Acetophenones & Derivatives (substituted);Building Blocks;C10;Carbonyl Compounds;Chemical Synthesis;Ketones;Organic Building Blocks | | Mol File: | 2161-85-5.mol | 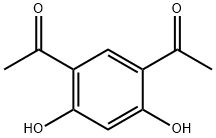 |
| | 4,6-DIACETYLRESORCINOL Chemical Properties |
| Melting point | 178-180 °C(lit.) | | Boiling point | 399.9±27.0 °C(Predicted) | | density | 1.300±0.06 g/cm3(Predicted) | | storage temp. | Inert atmosphere,Room Temperature | | pka | 7.87±0.23(Predicted) | | form | Crystalline Powder | | color | White to off-white | | λmax | 370nm(DMF)(lit.) | | CAS DataBase Reference | 2161-85-5(CAS DataBase Reference) |
| | 4,6-DIACETYLRESORCINOL Usage And Synthesis |
| Chemical Properties | Off-white powder | | Uses | 1,1′-(4,6-Dihydroxy-1,3-phenylene)bisethanone (4,6-diacetylresorcinol, DAR) may be used in the synthesis of the following:
- Schiff base ligands
- hexadentate chalcogenated bisimine ligands
- 1,5-benzodiazepines
- ketimine of chitosan
- mannich bases
- hydrazone ligands
- thiosemicarbazone, semicarbazone and thiocarbohydrazone ligands
- binuclear cobalt(II) and copper(II) complexes
- europium (III) complexes
| | Preparation | Preparation by Friedel–Crafts acylation of resorcinol,
? with acetic anhydride,
– in the presence of zinc chloride
at 142–150° for 15 min (96%) or at 150–160° for 20 min (68%)
; – in the presence of ferric chloride
. – in the presence of concentrated sulfuric acid at 130° for 15 min (15%)
; – in the presence of 70% perchloric acid at 125–135° for 15–20 min (42%)
. ? with acetyl chloride,
– in the presence of zinc chloride, at 120°
; – in the presence of ferric chloride, at 150° for 15 min (60%) or at reflux for 30 min
; – in the presence of concentrated sulfuric acid (18%)
. ? by a typical Friedel–Crafts reaction (24%). | | General Description | 1,1′-(4,6-Dihydroxy-1,3-phenylene)bisethanone (4,6-diacetylresorcinol, DAR) is a bifunctional carbonyl compound. Its synthesis by acetylating resorcinol in the presence of zinc chloride has been reported. The crystal structure of DAR has been studied. |
| | 4,6-DIACETYLRESORCINOL Preparation Products And Raw materials |
|