- 4,4'-Biphenol
-

- $6.00 / 1kg
-
2024-09-30
- CAS:92-88-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 2000KG/Month
- 4,4'-Biphenol
-

- $5.00 / 1KG
-
2024-09-29
- CAS:92-88-6
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10000kg
- 4,4'-Biphenol
-

- $0.00 / 1kg
-
2024-09-29
- CAS:92-88-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 1000000
|
| | 4,4'-Biphenol Basic information |
| Product Name: | 4,4'-Biphenol | | Synonyms: | (1,1’-Biphenyl)-4,4’-diol;BIPHENYL-4,4'-DIOL;4,4'-BIPHENOL;4,4'-BIPHENYLDIOL;1-(9,10-dihydroanthracen-1-yl)-9,10-dihydroanthracene;Biphenyl-4,4'-diol
4,4'-Dihydroxybiphenyl
4,4'-Dihydroxydiphenyl
4,4'-Diphenol;P,P'-BIPHENOL;P,P'-DIPHENOL | | CAS: | 92-88-6 | | MF: | C12H10O2 | | MW: | 186.21 | | EINECS: | 202-200-5 | | Product Categories: | Industrial/Fine Chemicals;Biphenyl derivatives;Biphenyl & Diphenyl ether;Phenoles and thiophenoles;Biphenyls (for High-Performance Polymer Research);Color Former & Related Compounds;Developer;Functional Materials;Reagent for High-Performance Polymer Research;bc0001 | | Mol File: | 92-88-6.mol | 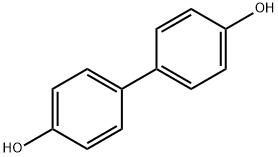 |
| | 4,4'-Biphenol Chemical Properties |
| Melting point | 280-282 °C(lit.) | | Boiling point | 280.69°C (rough estimate) | | density | 1.22 | | vapor pressure | 0Pa at 25℃ | | refractive index | 1.6010 (estimate) | | storage temp. | Sealed in dry,Room Temperature | | form | Powder | | pka | 9.74±0.26(Predicted) | | color | Off-white to light beige or light gray | | Water Solubility | sparingly soluble | | BRN | 1908886 | | LogP | 2.7 at 20℃ | | CAS DataBase Reference | 92-88-6(CAS DataBase Reference) | | NIST Chemistry Reference | p,p'-Biphenol(92-88-6) | | EPA Substance Registry System | 4,4'-Biphenyldiol (92-88-6) |
| Hazard Codes | Xn | | Risk Statements | 21-36/37/38-37/38/68-36 | | Safety Statements | 26-36-24/25-36/37 | | RIDADR | 3077 | | WGK Germany | 2 | | RTECS | DV4725000 | | TSCA | Yes | | HazardClass | 9 | | PackingGroup | III | | HS Code | 29061900 | | Toxicity | LD50 orl-rat: 9850 mg/kg TXAPA9 28,313,74 |
| | 4,4'-Biphenol Usage And Synthesis |
| Chemical Properties | Powder | | Uses | Biphenol is an intermediate used in the manufacture of thermoplastics such as liquid crystalline polymers (LCP), polyesters, polycarbonates and polysulfones. | | Uses | 4,4'-biphenol be used intermediate liquid crystal polymers raw materials. Synthetic polymers, due to its excellent heat resistance, it is used as polyester, urethane modified monomer, polycarbonate, polysulfone, and epoxy resin, for the manufacture of an excellent engineering plastics and composite materials. Antioxidant antioxidants for rubber and plastics. Dye intermediates or petroleum products stabilizer. | | Uses | Intermediates of Liquid Crystals | | Definition | ChEBI: A member of the class of hydroxybiphenyls that is biphenyl with hydroxy groups at positions 4 and 4'. | | Production Methods | 4,4'-Biphenol can be
synthesized in excellent yields by debutylation
of 3,3',5,5'-tetra-tert-butyl-4,4'-dihydroxybiphenyl. Other production processes are the
alkaline fusion of 4,4'-biphenyldisulfonic acid,
the boil down of benzidinebisdiazonium salts, and the decarboxylation of 4,4'-dihydroxybiphenyl-2-carboxylic acid. Symmetrically alkylated 4,4'-dihydroxybiphenyls such as 4,4'-dihydroxy-2,2',3,3',5,5',6,6'-hexamethylbiphenyl are preferably obtained by the
oxidative coupling of the corresponding substituted monophenols. Here, diphenoquinones are generally formed in the first step
which must be reduced. Phenol itself always
gives isomeric mixtures of dihydroxybiphenyls
on coupling with various oxidants. These
mixtures are also formed by alkaline fusion of
4,4'-dihydroxybiphenylsulfone. | | Flammability and Explosibility | Not classified | | Safety Profile | Poison by intraperitoneal route.Moderately toxic by skin contact. Mildly toxic byingestion. Human mutation data reported. When heated todecomposition it emits acrid smoke and irritating fumes. | | Purification Methods | Recrystallise the biphenol from aqueous EtOH preferably under N2 to avoid oxidation to the extended qiunone. It is characterized as the dimethyl derivative (4,4’-dimethoxybiphenyl) from which it is prepared by demethylation. The dimethoxy derivative has m 176.5-177o (from AcOH, EtOH, hexane or *C6H6) and sublimes in vacuo. [Williamson & Rodebush J Am Chem Soc 63 3019 1941, Beilstein 6 I 485, 6 II 962, 6 III 6389, 6 IV 6651.] |
| | 4,4'-Biphenol Preparation Products And Raw materials |
| Raw materials | 4,4'-Bis(trimethylsilyloxy)biphenyl-->6,6'-DI-TERT.BUTYL-4,4'-BI-ORTHO-CRESOL-->4-HYDROXY-4'-METHOXYBIPHENYL-->4,4'-Dimethoxybiphenyl-->3,3',5,5'-TETRA(TERT-BUTYL)[1,1'-BIPHENYL]-4,4'-DIOL-->4-Hydroxy-4'-fluorobiphenyl-->4,4'-Difluorobiphenyl-->1-Chloro-4-fluorobenzene-->4-Bromophenol | | Preparation Products | 4'-Hydroxy-4-biphenylcarbonitrile-->6fapbp-->3,3',5,5'-tetraformyl-4,4'-biphenyldiol-->4,4'-BIPHENYLDICARBONITRILE-->Cyanic acid, [1,1'-biphenyl]-4,4'-diyl ester-->[1,1'-biphenyl]-4,4'-diyl bis(2-Methylacrylate)-->3,3',5,5'-tetrabromo-[1,1'-biphenyl]-4,4'-diol |
|