|
|
| | COBALT ALUMINUM OXIDE Basic information |
| | COBALT ALUMINUM OXIDE Chemical Properties |
| | COBALT ALUMINUM OXIDE Usage And Synthesis |
| Chemical Properties | Cobalt aluminate is Not attacked by Cl2 , mineral acids or aqueous alkalies. Decomposed by fusion with KHSO4 and by heating with H2SO4 in a sealed tube at 200°C.
| | Uses | Cobalt Aluminum Oxide is used as a catalyst in nonoxidative propane dehydrogenation. | | Preparation |
A stoichiometric mixture of CoO and A12O3 is prepared and then mixed with 1.5 times its weight of KCl, The mixture is heated to about 1100°C in a porcelain crucible. The melt is cooled, pulverized, and extracted with boiling water until no further Cl- reaction is obtained. The residue is dried at 60°C in a drying oven.
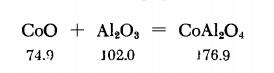
|
| | COBALT ALUMINUM OXIDE Preparation Products And Raw materials |
|