N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide manufacturers
|
| | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide Basic information |
| Product Name: | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide | | Synonyms: | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide;N-(8-amino-6-fluoro-5-methyl-1-oxo-3,4-dihydro-2H-naphthalen-2-yl)acetamide;N-(8-acetamido-6-fluoro-5-methyl-1-oxo-3,4-dihydro-2H-naphthalen-2-yl)acetamide;Acetamide, N-(8-amino-6-fluoro-1,2,3,4-tetrahydro-5-methyl-1-oxo-2-naphthalenyl)-;N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide hydrochloride;N-(8-amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalene-2-yl)acetamide;Exatecan Intermediate 2;Exatecan Impurity 2 | | CAS: | 182182-31-6 | | MF: | C13H15FN2O2 | | MW: | 250.27 | | EINECS: | | | Product Categories: | ADC;ADCs | | Mol File: | 182182-31-6.mol | 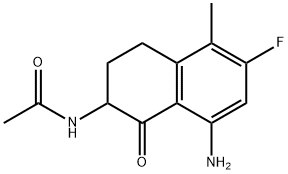 |
| | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide Chemical Properties |
| Boiling point | 550.1±50.0 °C(Predicted) | | density | 1.27±0.1 g/cm3(Predicted) | | storage temp. | Keep in dark place,Sealed in dry,2-8°C | | pka | 14.41±0.20(Predicted) | | form | Solid | | color | Off-white to gray | | InChI | InChI=1S/C13H15FN2O2/c1-6-8-3-4-11(16-7(2)17)13(18)12(8)10(15)5-9(6)14/h5,11H,3-4,15H2,1-2H3,(H,16,17) | | InChIKey | SEFPQWAVZCZXTK-UHFFFAOYSA-N | | SMILES | C(NC1CCC2=C(C1=O)C(N)=CC(F)=C2C)(=O)C |
| | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide Usage And Synthesis |
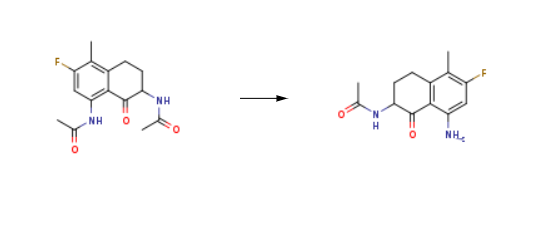
| Uses | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide is an important pharmaceutical raw material that is a by-product of the ezetimibe preparation process. | | Synthesis | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide is synthesised from N,N'-(3-fluoro-4-methyl-8-oxo-5,6,7,8-tetrahydronaphthalene-1,7-diyl)diethanamide by reaction in the presence of inert gas. The steps are as follows:
Add compound (15.5 g) to a solution of 2N hydrochloric acid (620 mL) and methanol (620 mL), protect with argon, and react at 60°C for 0.5 hours. The temperature is reduced to 010°C, and sodium carbonate (65.7 g ) In water (1314 mL) solution to pH=34, and then neutralized to pH=67 with saturated sodium bicarbonate solution, filtered, washed with 100 mL water, and dried to obtain N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide as a solid powder (12.4 g , 93%).
 |
| | N-(8-Amino-6-fluoro-5-methyl-1-oxo-1,2,3,4-tetrahydronaphthalen-2-yl)acetamide Preparation Products And Raw materials |
|