Mobocertinib succinate manufacturers
- Mobocertinib succinate
-
- $0.00 / 1kg
-
2024-07-17
- CAS:2389149-74-8
- Min. Order: 1kg
- Purity: >99.5% by HPLC
- Supply Ability: 100kg/month
- 788Succinic acid
-

- $90.00 / 1kg
-
2024-05-10
- CAS:2389149-74-8
- Min. Order: 10kg
- Purity: 0.99
- Supply Ability: 20tons
- Mobocertinib succinate
-

- $0.00 / 10g
-
2022-05-28
- CAS:2389149-74-8
- Min. Order: 1g
- Purity: 99%
- Supply Ability: 100KG
|
| | Mobocertinib succinate Basic information |
| Product Name: | Mobocertinib succinate | | Synonyms: | Mobocertinib succinate;788Succinic acid;TAK788 succinate;Mopotinib succinate;obocertinib Succinate (TAK-788 Succinate);Mobocertinib Succinate (TAK-788 Succinate);Mobocertinib succinate(TAK788);AP32788 succinate | | CAS: | 2389149-74-8 | | MF: | C36H45N7O8 | | MW: | 703.8 | | EINECS: | | | Product Categories: | API | | Mol File: | 2389149-74-8.mol |  |
| | Mobocertinib succinate Chemical Properties |
| storage temp. | Store at -20°C | | solubility | DMSO : 25 mg/mL (35.52 mM; Need ultrasonic) | | form | Solid | | color | White to yellow | | InChIKey | YXYAEUMTJQGKHS-UHFFFAOYSA-N | | SMILES | C(C(=O)O)CC(=O)O.C(C1=CN=C(NC2C=C(NC(=O)C=C)C(N(C)CCN(C)C)=CC=2OC)N=C1C1=CN(C)C2=CC=CC=C12)(=O)OC(C)C |
| | Mobocertinib succinate Usage And Synthesis |
| Description | Mobocertinib (TAK-788) succinate is an orally active and irreversible EGFR/HER2 inhibitor. Mobocertinib succinate potently inhibits oncogenic variants containing activating EGFRex20ins mutations with selectivity over wild-type EGFR. Mobocertinib succinate can be used in NSCLC research. | | Uses | Mobocertinib is an oral targeted therapy drug[1].Mobocertinib is used to treat a certain type of non-small cell lung cancer (NSCLC) that cannot be removed by surgery and has spread to other parts of the body either during or after treatment with platinum chemotherapy medications.
| | Mechanism of action | Mobocertinib acts to inhibit EGFR exon 20 insertion mutations at a lower concentration than it does on wild-type proteins. | | Side effects | Diarrhoea, rash, nausea, stomatitis, vomiting, decreased appetite, paronychia, fatigue, dry skin,musculoskeletal pain, increased amylase/lipase/creatinine, and decreased potassium/hemoglobin/magnesium/lymphocytes.
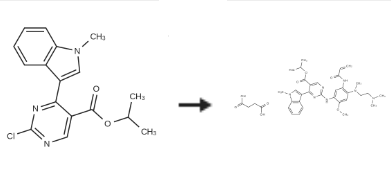
| | Synthesis | Mobocertinib succinate is prepared by the reaction of Isopropyl 2-chloro-4-(1-methyl-1H-indol-3-yl)pyrimidine-5-carboxylate in the presence of an inert atmosphere.
 | | References | [1] IMRANMOHD. Discovery, Development, Inventions, and Patent Trends on Mobocertinib Succinate: The First-in-Class Oral Treatment for NSCLC with EGFR Exon 20 Insertions.[J]. Biomedicines, 2021. DOI:10.3390/biomedicines9121938. |
| | Mobocertinib succinate Preparation Products And Raw materials |
|