|
|
| | CONGRESSANE Basic information |
| Product Name: | CONGRESSANE | | Synonyms: | PENTACYCLO[7.3.1.14,12.02,7.06,11]TETRADECANE;4,4'-Methylene-1,1'-methylene-5,6'-cyclo-3,2'-bicyclohexane;Pentacyclo[5.3.1.12,6.14,11.19,14]tetradecane;Congressane
Pentacyclo[7.3.1.1(4,12).0(2,7).0(6,11)]tetradecane;DIAMANTANE;DIAMANTINE;CONGRESSANE;Diamantane> | | CAS: | 2292-79-7 | | MF: | C14H20 | | MW: | 188.31 | | EINECS: | | | Product Categories: | Adamantane derivatives | | Mol File: | 2292-79-7.mol | 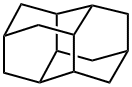 |
| | CONGRESSANE Chemical Properties |
| Melting point | 236-237° | | Boiling point | 258.34°C (rough estimate) | | density | 1.092 | | refractive index | 1.5500 (estimate) | | Fp | 98.1℃ | | form | powder to crystal | | color | White to Light yellow | | Merck | 14,2538 |
| | CONGRESSANE Usage And Synthesis |
| Definition | ChEBI: Diamantane is a polycyclic cage and a polycyclic alkane. | | Purification Methods | Purify diamantane by repeated crystallisation from MeOH or pentane. Alternatively purify it by dissolving it in CH2Cl2, washing with 5% aqueous NaOH and water, and drying (MgSO4). The solution is filtered, concentrated to a small volume, an equal weight of alumina is added, and the solvent evaporated. The residue is placed on an activated alumina column (ca 4 x weight of diamantane) and eluted with pet ether (b 40-60o). Eight sublimations and twenty zone refining experiments gave material m 251o of 99.99% purity by differential analysis [Gund et al. Tetrahedron Lett 3877 1970, Courtney et al. J Chem Soc Perkin Trans I 2691 1972]. [For spectra see Cupas et al. J Am Chem Soc 87 919 1965.] |
| | CONGRESSANE Preparation Products And Raw materials |
|