|
|
| | (R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DM-SEGPHOS Basic information | | Reaction |
| Product Name: | (R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DM-SEGPHOS | | Synonyms: | (R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DM-SEGPHOS;(R)-DM-SEGPHOS(R);(R)-(+)-DM-SEGPHOS(regR);(R)-(+)-DM-SEGPHOS®;(R)-DM-SEGPHOS;(R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole;(R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole, min. 98% (R)-(+)-DM-SEGPHOS(R);1,1'-[(4R)-[4,4'-Bi-1,3-benzodioxole]-5,5'-diyl]bis[1,1-bis(3,5-dimethylphenyl)phosphine] | | CAS: | 850253-53-1 | | MF: | C46H44O4P2 | | MW: | 722.8 | | EINECS: | | | Product Categories: | | | Mol File: | 850253-53-1.mol | ![(R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DM-SEGPHOS Structure](CAS/20180808/GIF/850253-53-1.gif) |
| | (R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DM-SEGPHOS Chemical Properties |
| Melting point | 256-261 °C | | Boiling point | 816.9±65.0 °C(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | form | Powder | | color | off white to pale yellow | | optical activity | [α]20/D +60, c = 0.1 in chloroform |
| WGK Germany | 3 | | HS Code | 2932.99.7000 |
| | (R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DM-SEGPHOS Usage And Synthesis |
| Reaction |
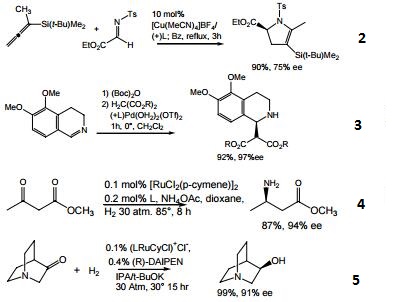
- Biaryl bisphosphine ligand with narrow dihedral angle. The DM-SEGPHOS ligand, as the ruthenium complex, gives superior enantioselectivity and diastereoselectivity in the asymmetric hydrogenation of α-substituted-β-ketoesters.
- Copper catalyzed enantioselective [3 + 2] cycloaddition as a route to γ–amino ketones and 3-pyrrolidinones.
- Palladium catalyzed enantioselective addition of malonates to dihydroisoquinolines.
- Ruthenium catalyzed enantioselective synthesis of β amino acids by hydrogenation.
- Ruthenium catalyzed asymmetric hydrogenation of 3-quinuclidinone. See 44-0098 for Ru catalyst.
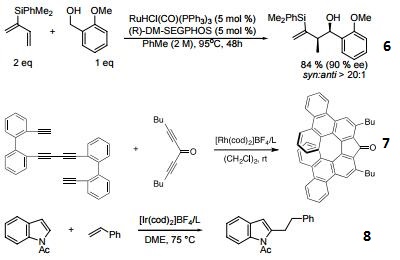
- Diastereoand enantioselective ruthenium-catalyzed hydrohydroxyalkylation of 2-silyl-butadienes.
- Asymmetric [2+2+2] cycloaddition.
- Linear selective C-H activation.
| | Uses | Takasago Ligands and Complexes for Asymmetric Reactions |
| | (R)-(+)-5,5'-Bis[di(3,5-xylyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DM-SEGPHOS Preparation Products And Raw materials |
|