- Bifonazole
-

- $45.00 / 1kg
-
2025-04-17
- CAS:60628-96-8
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 20 tons
- Bifonazole
-

- $0.00 / 25Kg/Drum
-
2025-04-16
- CAS:60628-96-8
- Min. Order: 1KG
- Purity: 98%-100.5%
- Supply Ability: 1000kg
- Bifonazole
-

- $10.00 / 1KG
-
2025-04-16
- CAS:60628-96-8
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 100 mt
Related articles - What is bifonazole used for?
- Bifonazoleis a imidazole antimycotic bifonazole used to treat mycoses of the skin induced or sustained by fungi such as dermat....
- Mar 16,2024
|
| | Bifonazole Basic information |
| Product Name: | Bifonazole | | Synonyms: | Bifonazole for system suitability;Bifonazole Impurity;Biphenylylbenzylazole;trifonazole;1-[P,ALPHA-DIPHENYLBENZYL]IMIDAZOLE;1[4,ALPHA-DIPHENYLBENZYL]-IMIDAZOLE;BIFONAZOLE;1-(p,α-diphenylbenzyl)imidazole | | CAS: | 60628-96-8 | | MF: | C22H18N2 | | MW: | 310.4 | | EINECS: | 262-336-6 | | Product Categories: | API;SARAFLOX | | Mol File: | 60628-96-8.mol | 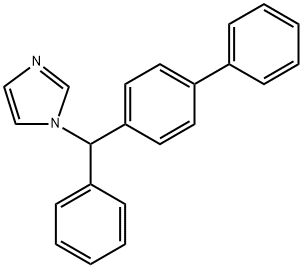 |
| | Bifonazole Chemical Properties |
| Melting point | 142℃ | | Boiling point | 440.55°C (rough estimate) | | density | 1.1150 (rough estimate) | | refractive index | 1.7620 (estimate) | | storage temp. | Sealed in dry,Room Temperature | | solubility | Soluble in DMSO | | form | powder | | pka | 6.55±0.22(Predicted) | | color | white to off-white | | Merck | 14,1213 | | LogP | 4.770 | | CAS DataBase Reference | 60628-96-8(CAS DataBase Reference) |
| Hazard Codes | Xn | | Risk Statements | 22 | | WGK Germany | 1 | | RTECS | NI3517000 | | HS Code | 29332900 | | Toxicity | LD50 in male mice, rats (mg/kg): 2629, 2854 orally (Schlüter) |
| | Bifonazole Usage And Synthesis |
| Description | Bifonazole represents the first topical broad spectrum antimycotic approved for
once daily administration. Its in vitro activity appears equivalent to its structural
relative clotrimazole, being effective against dermatophytes, other filamentous
fungi, dimorphic fungi and yeasts. | | Description | Bifonazole is a topically-active imidazole antifungal compound that has broad spectrum activity in vitro against dermatophytes, molds, yeasts, dimorphic fungi, and some Gram-positive bacteria. It is effective in the treatment of experimental dermatophytic and Candida in animals. Bifonazole is also a potent inhibitor of cytochrome P450 aromatase (Ki = 68 nM, IC50 = 270 nM), which catalyzes the biosynthesis of estrogens from androgens. When applied topically in animals, it demonstrates prolonged retention time in skin with minimal percutaneous absorption, thus minimizing its effect on aromatase. | | Chemical Properties | White or almost white, crystalline powder. | | Originator | Bayer (W. Germany) | | Uses | antibacterial | | Uses | Bifonazole is an imidazole antifungal agent and calmodulin antagonist. It causes a reduction in glycolysis and ATP levels in B16 melanoma cells. Bifonazole is an inhibitor of C14orf1. Bifonazole is an imidazole-based anti-fungal agent with broad spectrum activity against many fungi, molds, yeast and some gram-positive bacteria. Bifonazole inhibits ergosterol biosynthetic protein 28 and Cytochrome P450 2B4. | | Definition | ChEBI: 1-[biphenyl-4-yl(phenyl)methyl]imidazole is a member of the class of imidazoles carrying an alpha-(biphenyl-4-yl)benzyl substituent at position 1. It is a member of imidazoles and a member of biphenyls. | | Manufacturing Process | 38.8g (0.15 mol) of 4-phenylbenzophenone are dissolved in 200 ml of ethanol
and 39 (0.075 mol) of sodium borohydride are added. After heating for 15
hours under reflux, and allowing to cool, the reaction mixture is hydrolyzed
with water containing a little hydrochloric acid. The solid thereby produced is
purified by recrystallization from ethanol. 36 g (89% of theory) of (biphenyl-
4-yl)-phenyl-carbinol [alternatively named as diphenyl-phenyl carbinol or α-
(biphenyl-4-yl)benzylalcohol] of melting point 72°-73°C are obtained.
13.6 g (0.2 mol) of imidazole are dissolved in 150 ml of acetonitrile and 3.5
ml of thionyl chloride are added at 10°C. 13 g (0.05 mol) of (biphenyl-4-yl)-
phenyl-carbinol are added to the solution of thionyl-bis-imidazole thus
obtained. After standing for 15 hours at room temperature, the solvent is
removed by distillation in vacuo. The residue is taken up in chloroform and
the solution is washed with water. The organic phase is collected, dried over
sodium sulfate and filtered and the solvent is distilled off in vacuo. The oily
residue is dissolved in ethyl acetate and freed from insoluble, resinous
constituents by filtration. The solvent is again distilled off in vacuo and the
residue is purified by recrystallization from acetonitrile, 8.7 g (56% of theory)
of (biphenyl-4-yl)-imidazol-1-yl-phenylmethane [alternatively named as
diphenyl-imidazolyl-(1)-phenyl-methane or as 1-(α-biphenyl-4-
ylbenzyl)imidazole] of melting point 142°C are obtained. | | Brand name | Mycospor
(Bayer). | | Therapeutic Function | Antifungal |
| | Bifonazole Preparation Products And Raw materials |
|