|
|
| | 1-Bromo-2,3-difluorobenzene Basic information |
| Product Name: | 1-Bromo-2,3-difluorobenzene | | Synonyms: | 2,3- twofluorine broMine;1-Bromo-2,3-difluorobenzene 98%;1-BROMO-2,3-DIFLUOROBENZENE;1-Bromo-2,3-Difluorobenzenne;2,3-Difluorobromobenzene 98%;2,3-Difluorobromobenzene98%;1-Bromo-2,3-difluorobenzene,98%;1-Bromo-2,3-difluoro | | CAS: | 38573-88-5 | | MF: | C6H3BrF2 | | MW: | 192.99 | | EINECS: | 609-564-8 | | Product Categories: | Miscellaneous;Aryl;C6;Halogenated Hydrocarbons;Fluorobenzene Series;Fluorobenzene;Aromatic Halides (substituted);Other fluorin-contained compounds;Purines;38573-88-5 | | Mol File: | 38573-88-5.mol | 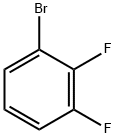 |
| | 1-Bromo-2,3-difluorobenzene Chemical Properties |
| Boiling point | 234 °C (765 mmHg) | | density | 1.724 g/mL at 25 °C(lit.) | | refractive index | n20/D 1.509(lit.) | | Fp | 142 °F | | storage temp. | 2-8°C | | form | clear liquid | | color | Colorless to Almost colorless | | Specific Gravity | 1.724 | | InChIKey | RKWWASUTWAFKHA-UHFFFAOYSA-N | | CAS DataBase Reference | 38573-88-5(CAS DataBase Reference) |
| Hazard Codes | Xi,F | | Risk Statements | 36/37/38 | | Safety Statements | 26-36/37/39-37/39 | | RIDADR | 1993 | | WGK Germany | 3 | | Hazard Note | Flammable | | HazardClass | 3.2 | | PackingGroup | III | | HS Code | 29039990 |
| | 1-Bromo-2,3-difluorobenzene Usage And Synthesis |
| Chemical Properties | Clear colorless to peach liquid | | Uses | 1-Bromo-2,3-difluorobenzene may be used in the synthesis of potent, orally active Calcitonin gene-related peptide (CGRP) receptor antagonist (BMS-846372). It may be used in the preparation of 2,3-difluorophenyl(dimethyl)phosphane. | | Uses | intermediate for liquid crystal and drugs | | General Description | Microbial oxidation of 1-bromo-2,3-difluorobenzene by Pseudomonas putida strain 39/D and Escherichia coli recombinant microorganism (strain JM 109(pDTG601)) has been reported. | | Synthesis | Add
50% aqueous solution of KOH (18.0 g, 160 mmol) to a mixture of
1-bromo-5,5,6,6-tetrafluorocyclohex-1-ene (11.65 g) and
triethylbenzylammonium chloride (0.15 g, 0.7 mmol) at 30-35°C for 30
minutes. Keep the reaction mixture at 75-85°C for 2 hours. Cool the
reaction mixture. Dilute the reaction mixture with water. Extract the
organic product with CH2Cl2. Dry the organic product over CaCl2. Distill the organic product. 1H NMR (CDCl3, 300.1 MHz), δ: 7.00-7.16 (m, 1H, Ar); 7.17-7.29 (m, 1H, Ar); 7.34-7.47 (m, 1H, Ar). 13C NMR (CDCl3,
75.5 MHz), δ: 110.40 (d, C(1), J = 17.5 Hz); 116.40 (d, C(4), J = 17.7
Hz); 124.70 (dd, C(5), J = 7.1 Hz, J = 5.0 Hz); 128.23 (d, C(6), J = 3.6
Hz); 148.10 (dd, C(2), J = 248.8 Hz, J = 14.3 Hz); 150.92 (dd, C(3), J =
251.9 Hz, J = 13.3 Hz). 19F NMR (CDCl3, 282.4 MHz), δ: -130.9 (m, 1 F, Ar), -134.8 (m, 1 F, Ar). BP 157-158°C. Elemental Analysis Found (%): C, 37.54; H, 1.50. C6H3BrF2. Calculated (%): C, 37.34; H, 1.52. Mass Spec MS, m/z (Irel (%)): 194, 192 [M]+ (100, 99), 113 [M-Br]+ (88), 63 (60).
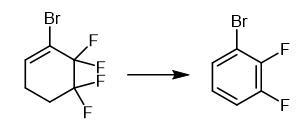 Fig The synthetic method of 1-Bromo-2,3-difluorobenzene Fig The synthetic method of 1-Bromo-2,3-difluorobenzene | | storage | Keep container sealed and store in an ventilated, low temperature, dry warehouse, separate from foods and oxidizing agents. |
| | 1-Bromo-2,3-difluorobenzene Preparation Products And Raw materials |
|