- Erythromycin
-

- $1.00 / 1kg
-
2025-04-18
- CAS:114-07-8
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10 mt
- Erythromycin
-

- $0.00 / 1Kg/Bag
-
2025-04-18
- CAS:114-07-8
- Min. Order: 1Kg/Bag
- Purity: 0.99
- Supply Ability: 20 tons
- Erythromycin
-

- $0.00 / 25Kg/Drum
-
2025-04-16
- CAS:114-07-8
- Min. Order: 1KG
- Purity: 93%-102%; EP
- Supply Ability: 500KGS
Related articles - What is Erythromycin?
- Erythromycin (CAS: 114-07-8), thereafter renamed erythromycin A, was first isolated at Eli Lilly from a strain of Streptomyces....
- Mar 15,2022
|
| | Erythromycin Basic information |
| | Erythromycin Chemical Properties |
| Melting point | 133 °C | | alpha | -74.5 º (c=2, ethanol) | | Boiling point | 719.69°C (rough estimate) | | density | 1.1436 (rough estimate) | | refractive index | -74 ° (C=2, EtOH) | | storage temp. | Inert atmosphere,Room Temperature | | solubility | ethanol: soluble | | form | powder | | pka | 8.8(at 25℃) | | color | white to faint yellow | | biological source | Streptomyces erythreus | | optical activity | [α]/D -78 to --71° | | Water Solubility | Soluble in water at 2mg/ml | | Merck | 14,3681 | | BRN | 8183758 | | InChIKey | HIYRERIGRWIODP-PNFGZDISSA-N | | EPA Substance Registry System | Erythromycin (114-07-8) |
| | Erythromycin Usage And Synthesis |
| Description | Erythromycin ethyl succinate is a mixed double ester pro-drug in which one carboxyl of succinic acid
esterifies the C-2′ hydroxyl of erythromycin and the other ethanol. This pro-drug frequently is
used in an oral suspension for pediatric use largely to mask the bitter taste of the drug. Film-coated tablets
also are used to deal with this. Some cholestatic jaundice is associated with the use of EES. | | Description | Erythromycin is a macrolide antibiotic that inhibits bacterial protein synthesis by targeting the 50S ribosomal subunit, blocking the progression of nascent polypeptide chains. It is active against a host of bacterial genera, including Streptococcus, Staphylococcus, and Haemophilus (MIC90s = 0.015-2.0 mg/l). Erythromycin (10-40 mg/kg) dose-dependently inhibits the growth of S. aureus in a mouse model of thigh infection. It also inhibits the cytochrome P450 (CYP450) isoform CYP3A4 in vitro with IC50 values of 33 and 27.3 μM for α-hydroxytriazolam and 4-hydroxytriazolam formation, respectively, following administration of triazolam, which is known to be metabolized primarily by CYP3A4. Formulations containing erythromycin have been used in the treatment of bacterial respiratory and skin infections, pertussis, and a variety of other bacterial infections. | | Chemical Properties | White to off white crystalline powder | | Originator | Ilotycin,Dista,US,1952 | | Uses | Macrolide antibacterial | | Uses | Labeled Erythromycin, intended for use as an internal standard for the quantification of Erythromycin by GC- or LC-mass spectrometry. | | Uses | Erythromycin A is a 14-membered macrocyclic lactone with broad spectrum antibiotic activity, isolated from Saccharopolyspora erythraea (formerly Streptomyces erythreus) in 1952. Erythromycin is one of only a handful of microbial metabolites to have profoundly shaped the treatment of bacterial disease in the last 50 years. Erythromycin has given rise to new generations of semi-synthetic derivatives with improved stability and potency. Our product has been HPLC-purified to remove contaminants and degradation products. | | Uses | For use in the treatment of infections caused by susceptible strains of microorganisms in the following diseases: respiratory tract infections (upper and lower) of mild to moderate degree, pertussis (whooping cough), as adjunct to antitoxin in infections | | Definition | An antibiotic produced by growth of Streptomyces
erythreus Waksman. It is effective against infections caused by Gram-positive bacteria, including
some β-hemolytic streptococci, pneumococci, and
staphylococci. | | Indications | Erythromycin is an antibiotic in the macrolide family
that also has promotility effects because
it is a motilin agonist. | | Manufacturing Process | An inoculum broth is prepared having the following composition: 32 pounds
starch; 32 pounds soybean meal; 10 pounds corn steep solids; 10 pounds
sodium chloride; 6 pounds calcium carbonate; and 250 gallons water.
The broth is placed in an iron tank of 350 gallon capacity and is sterilized by
heating it under pressure at a temperature of about 120°C for 30 minutes.
The sterilized broth is cooled and inoculated aseptically with spores of
Streptomyces erythreus, NRRL 2338. The organism is grown in the broth at
about 26°C for a period of 45 hours. During the growth period the broth isstirred and aerated with sterile air in the amount of about 0.5 volume of air
per volume of culture broth per minute.
In a 1,600-gallon iron tank is placed a fermentation broth having the following
composition: 153 pounds starch; 153 pounds soybean meal; 51 pounds corn
steep solids; 33 pounds calcium carbonate; 51 pounds sodium chloride; and
1,200 gallons water.
The culture broth is sterilized by heating it under pressure at about 120°C for
about 30 minutes. The broth is cooled and the above inoculant culture is
added aseptically. The organism is grown in the broth for 4 days at a
temperature of 26°C. During the growth period the broth is stirred and sterile
air is blown through the broth at a rate of about 0.5 volume of air per volume
of broth per minute. At the end of the growth period the broth shows an
antibiotic activity equivalent to about 150 mcg of erythromycin per ml of
broth.
The culture broth (about 1,100 gallons in volume) is adjusted to pH 9.5 with
40% sodium hydroxide solution and is filtered to remove the mycelium, the
filtration being assisted by use of 3% of Hyflo Super-Cel, a filter aid, (sold by
Johns-Manville Company). The clear filtrate is extracted with amyl acetate in a
Podbielniak extractor using a ratio of 1 volume of amyl acetate to 6 volumes
of clarified broth. The amyl acetate extract is in turn extracted batchwise with
water brought to about pH 5 by the addition of sulfuric acid. Two extractions
are carried out, the first with ? volume and the second with ? volume of
water adjusted to pH 5 with sulfuric acid. The aqueous extracts are combined
and adjusted to pH 8.0 with sodium hydroxide solution.
The alkaline solution is concentrated in vacuo to a volume of about 30 gallons
and the solution is then adjusted to pH 9.5 by the addition of aqueous sodium
hydroxide and is allowed to stand. Erythromycin separates as a crystalline
material. The crystals are filtered off, the mother liquor is adjusted to about
pH 8 by the addition of dilute sulfuric acid and is concentrated in vacuo to a
volume of about 30 gallons. The solution is adjusted to about pH 9.5 and
allowed to stand, whereupon an additional amount of erythromycin separates
in crystalline form. The total amount of erythromycin obtained is about 256
grams. The erythromycin is purified by several recrystallizations from aqueous
acetone (2:1 mixture), according to US Patent 2,653,899. | | Brand name | Ilotycin (Dista). | | Therapeutic Function | Antibacterial | | Antimicrobial activity | Gram-positive rods, including Clostridium spp. (MIC50 0.1–1 mg/L), C. diphtheriae (MIC50 0.1–1 mg/L), L. monocytogenes (MIC50 0.1–0.3 mg/L) and Bacillus anthracis (MIC50 0.5–1.0 mg/L), are generally susceptible. Most strains of M. scrofulaceum and M. kansasii are susceptible (MIC50 0.5–2 mg/L), but M. intracellulare is often and M. fortuitum regularly resistant. Nocardia isolates are resistant. H. ducreyi, B. pertussis (MIC50 0.03–0.25 mg/L), some Brucella, Flavobacterium, Legionella (MIC50 0.1–0.5 mg/L) and Pasteurella spp. are susceptible. H. pylori (MIC 0.06–0.25 mg/L) and C. jejuni are usually susceptible, but C. coli may be resistant. Most anaerobic bacteria, including Actinomyces and Arachnia spp., are susceptible or moderately so, but B. fragilis and Fusobacterium spp. are resistant. T. pallidum and Borrelia spp. are susceptible, as are Chlamydia spp. (MIC ≤0.25 mg/L), M. pneumoniae and Rickettsia spp. M. hominis and Ureaplasma spp. are resistant.
Enterobacteriaceae are usually resistant. Activity rises with increasing pH up to 8.5. Incubation in 5–6% CO2 raises the MIC for H. influenzae from 0.5–8 to 4–32 mg/L; MICs for Str. pneumoniae and Str. pyogenes also rise steeply. Activity is predominantly bacteristatic. | | Acquired resistance | In Europe, the USA and other countries the incidence of resistance in Str. pneumoniae ranges from 5% to over 60%. In Str. pneumoniae strains resistant or intermediately susceptible to penicillin G, resistance rates above 80% have been reported. Increasing rates of resistance in clinical isolates of Str. pyogenes have also been reported, threatening its use as an alternative to penicillin G in allergic patients.
Lower rates of resistance have been reported in other bacterial species, including methicillin-resistant Staph. aureus, coagulase-negative staphylococci, Str. agalactiae, Lancefield group C and G streptococci, viridans group streptococci, H. pylori, T. pallidum, C. diphtheriae and N. gonorrhoeae. | | General Description | Early in 1952, McGuire et al. reported the isolation oferythromycin (E-Mycin, Erythrocin, Ilotycin) fromStreptomyces erythraeus. It achieved rapid early acceptanceas a well-tolerated antibiotic of value for the treatment ofvarious upper respiratory and soft-tissue infections causedby Gram-positive bacteria. It is also effective against manyvenereal diseases, including gonorrhea and syphilis, andprovides a useful alternative for the treatment of many infectionsin patients allergic to penicillins. More recently,erythromycin was shown to be effective therapy for Eatonagent pneumonia (Mycoplasma pneumoniae), venereal diseasescaused by Chlamydia, bacterial enteritis caused byCampylobacter jejuni, and Legionnaires disease.
The commercial product is erythromycin A, whichdiffers from its biosynthetic precursor, erythromycin B,in having a hydroxyl group at the 12-position of theaglycone. The chemical structure of erythromycin A was reportedby Wiley et al.197 in 1957 and its stereochemistry byCelmer198 in 1965. An elegant synthesis of erythronolide A,the aglycone present in erythromycin A, was described byCorey et al.
The amino sugar attached through a glycosidic link to C-5 is desosamine, a structure found in several other macrolideantibiotics. The tertiary amine of desosamine (3,4,6-trideoxy-3-dimethylamino-D-xylo-hexose) confers a basiccharacter to erythromycin and provides the means by whichacid salts may be prepared. The other carbohydrate structurelinked as a glycoside to C-3 is called cladinose (2,3,6-trideoxy-3-methoxy-3-C-methyl-L-ribo-hexose) and isunique to the erythromycin molecule. | | Pharmaceutical Applications | A natural antibiotic produced as a complex of six components (A–F) by Saccharopolyspora erythraea. Only erythromycin A has been developed for clinical use. It is available in a large number of forms for oral administration: the base compound (enteric- or film-coated to prevent destruction by gastric acidity); 2′-propionate and 2′-ethylsuccinate esters; a stearate salt; estolate and acistrate salts of 2′-esters. The 2′-esters and their salts have improved pharmacokinetic and pharmaceutical properties and are less bitter than erythromycin. It is also formulated as the lactobionate and gluceptate forparenteral use. | | Biological Activity | The oral bioavailability of erythromycin base is poor and is highly
variable because of inactivation by gastric acidity. Formulations with an acid-resistant coating have therefore been
developed, as well as esters with improved oral bioavailability. Stearate
is hydrolyzed in the intestine, whereas ethylsuccinate is absorbed both
as the free base (55%) and the ester (45%) formulations. These are best
absorbed in the fasting state. Estolate absorption is not affected by food;
20–30% of the serum concentration corresponds to the active form and
70–80% to the ester prodrug.
Serum protein binding varies between 40% and 90%. Alcohol can cause
a moderate reduction in the absorption of erythromycin succinate. | | Biological Activity | Erythromycin is the principal one in antimicrobial drugs. Although available as the parent entity, semisynthetic derivatives have proved to be clinically superior to the natural cogener. Like the tetracyclines, synthetic transformations in the macrolide series have not significantly altered their antibacterial spectra, but have improved the pharmacodynamic properties. For example, the propionate ester of erythromycin lauryl sulfate (erythromycin estolate) has shown greater acid stability than the unesterified parent substance. Although the estolate appears in the blood somewhat more slowly, the peak serum levels reached are higher and persist longer than other forms of the drug. However, cholestatic hepatitis may occasionally follow administration of the estolate and, for that reason, the stearate is often preferred. Erythromycin is effective against Group A and other nonenterococcal streptococci, Corynebacterium diphtheriae, Legionella pneumophila, Chlamydia trachomatis, Mycoplasma pneumoniae, and Flavobacterium. Because of the extensive use of erythromycin in hospitals, a number of Staph. aureus strains have become highly resistant to the drug. For this reason, erythromycin has been used in combination with chloramphenicol. This combination is also used in the treatment of severe sepsis when etiology is unknown and patient is allergic to penicillin. | | Biochem/physiol Actions | Mode of Action: Erythromycin acts by inhibiting elongation at the transpeptidation step, specifically aminoacyl translocation from the A-site to P-site by binding to the 50s subunit of the bacterial 70s rRNA complex.Antimicrobial Spectrum: This product acts against both gram-negative and gram-positive bacteria. | | Mechanism of action | Macrolides are inhibitors of protein synthesis at the ribosomes. They impair the elongation cycle of the
peptidyl chain by specifically binding to the 50S subunit of the
ribosome. Specificity toward prokaryotes relies upon the absence of
50S ribosomes in eukaryotes. The main interaction site is located at
the central loop of the domain V of the 23S rRNA, at the vicinity
of the peptidyl transferase center. The macrolide binding site is located
at the entrance of the exit tunnel used by the nascent peptide chain to
escape from the ribosome, at the place where the central loop of
domain V interacts with proteins L4 and L22 and with the loop of
754 Macrolides and Ketolides
hairpin 35 in domain II of rRNA.
Interaction occurs via the formation of hydrogen-bonds between the
reactive groups (2u-OH) of the desosamine sugar and the lactone ring and adenine residue 2058. This explains why
mutation or methylation in position 2058 as well as mutations in proteins
L4 and L22 confer resistance to macrolides. The binding site of macrolides on the ribosome overlaps that of chloramphenicol or lincosamides
such as clindamycin, explaining pharmacologic
antagonism between these antibiotic classes as well as cross-resistance. | | Pharmacology | Erythromycin inhibits bacterial protein synthesis by reversibly binding with their 50
S ribosomal subunit, thus blocking the formation of new peptide bonds. Erythromycin is
classified as a bacteriostatic antibiotic.
However, it can also exhibit a bactericidal effect against a few types of microbes at cer�tain concentrations.
Bacterial resistance to erythromycin can originate by two possible mechanisms: the
inability of reaching the cell membrane, which is particularly relevant in the case of the
microorganisms Enterobacteriaceae, or in the case of the presence of a methylated alanine
in the 23 S ribosomal RNA of the 50 S subunit, which lowers the affinity of erythro�mycin to it.
Erythromycin acts on Gram-positive (staphylococci both produced and not produced by
penicillinase, streptococci, pneumococci, clostridia) and a few Gram-negative microorgan�isms (gonococci, brucelli, hemophile and whooping cough bacilli, legionelli), mycoplasma,
chlamydia, spirochaeta, and Rickettsia. Colon and blue-pus bacilli, as well as the bacilli
shigella, salmonella, and others are resistant to erythromycin. | | Pharmacokinetics | absorption and metabolism
The acid lability of erythromycin base necessitates administration in a form giving protection from gastric acid. In acid media it is rapidly degraded (10% loss of activity at pH 2 in less than 4 s) by intramolecular dehydrogenation to a hemiketal and hence to anhydroerythromycin A, neither of which exerts antibacterial activity. Delayed and incomplete absorption is obtained from coated tablets and there is important inter- and intra-individual variation, adequate levels not being attained at all in a few subjects. Food delays absorption of erythromycin base. After 500 mg of the 2′-ethylsuccinyl ester, mean peak plasma levels at 1–2 h were 1.5 mg/L. In subjects given 1 g of the 2′-ethylsuccinate every 12 h for seven doses, the mean plasma concentration 1 h after the last dose was around 1.4 mg/L. Intra- and inter-subject variation and delayed and erratic absorption in the presence of food have not yet been eliminated by new formulations. Improved 500 mg preparations of erythromycin stearate are claimed to produce peak plasma levels of 0.9–2.4 mg/L that are little affected by the presence of food. 2′-Esters of erythromycin are partially hydrolyzed to erythromycin: 2′-acetyl erythromycin is hydrolyzed more rapidly than the 2′-propionyl ester, but more slowly than the 2′-ethylsuccinate.
The stoichiometric mixture with stearate does not adequately protect erythromycin from acid degradation. After an oral dose of erythromycin stearate, equivalent concentrations of erythromycin and its main degradation product, anhydroerythromycin, could be detected.
Doses of 10 mg/kg produced mean peak plasma concentrations around 1.8 mg/L in infants weighing 1.5–2 kg and 1.2 mg/L in those weighing 2–2.5 kg. In infants less than 4 months old, doses of 10 mg/kg of the 2′-ethylsuccinate every 6 h produced steady state plasma levels of around 1.3 mg/L. The apparent elimination half-life was 2.5 h. In children given 12.5 mg/kg of erythromycin 2′-ethylsuccinate every 6 h, the concentration in the plasma 2 h after the fourth dose was around 0.5–2.5 mg/L.
Distribution
Very low levels are obtained in cerebrospinal fluid (CSF), even in the presence of meningeal inflammation, and after parenteral administration. Levels of 0.1 mg/L in aqueous humor were found when the serum level was 0.36 mg/L, but there was no penetration into the vitreous. In children with otitis media given 12.5 mg/kg of erythromycin 2′- ethylsuccinate every 6 h, concentrations in middle ear exudate were 0.25–1 mg/L. In patients with chronic serous otitis media given 12.5 mg/kg up to a maximum of the equivalent of 500 mg, none was detected in middle ear fluid, but on continued treatment levels up to 1.2 mg/L have been described.
Penetration also occurs into peritoneal and pleural exudates. Mean concentrations of 2.6 mg/L have been found in sputum in patients receiving 1 g of erythromycin lactobionate intravenously every 12 h and 0.2–2 mg/L in those receiving an oral stearate formulation. Levels in prostatic fluid are about 40% of those in the plasma. Salivary levels of around 4 mg/L were found in subjects receiving doses of 0.5 g every 8 h at 5 h after a dose, when the plasma concentration was around 5.5 mg/L. Intracellular:extracellular ratios of 4–18 have been found in polymorphonuclear neutrophils.
Fetal tissue levels are considerably higher after multiple doses: when the mean peak maternal serum level was 4.94 (0.66–8) mg/L, the mean fetal blood concentration was 0.06 (0–0.12) mg/L. Concentrations were more than 0.3 mg/L in amniotic fluid and most other fetal tissues, but the concentrations were variable and unmeasurable in some. Erythromycin appears to be concentrated by fetal liver.
excretion
Erythromycin is excreted both in urine and in the bile but only a fraction of the dose can be accounted for in this way. Only about 2.5% of an oral dose or 15% of an intravenous dose is recovered unchanged in the urine. It is not removed to any significant extent by peritoneal dialysis or hemodialysis. Reported changes in apparent elimination half-life in renal impairment may be related to the saturable nature of protein binding. Fairly high concentrations (50–250 mg/L) are found in the bile. In cirrhotic patients receiving 500 mg of the base, peak plasma levels were higher and earlier than in healthy volunteers (2.0 and 1.5 mg/L
at 4.6 and 6.3 h, respectively). The apparent elimination half-life was 6.6 h. It is possible that the smaller excretion of the 2′-propionyl ester in the bile in comparison to the base accounts in part for its better-maintained serum levels. There is some enterohepatic recycling, but some of the administered dose is lost in the feces, producing concentrations of around 0.5 mg/g. | | Clinical Use | Erythromycin is used (offlabel
indication) to accelerate gastric emptying in diabetic
gastroparesis and postoperative gastroparesis.
Tachyphylaxis will occur, so it cannot be used uninterruptedly
for long periods. | | Side effects | Oral administration, especially of large doses, commonly causes epigastric distress, nausea and vomiting, which may be severe. Solutions are very irritant: intravenous infusions almost invariably produce thrombophlebitis. Cholestatic hepatitis occurs rarely. Transient auditory disturbances have been described after intravenous administration of the lactobionate salt, and occasionally in patients with renal and hepatic impairment in whom oral dosage has produced high plasma levels. Sensorineural hearing impairment can occur and, although this is usually a reversible effect which occurs at high dosage, can be permanent. Prolongation of the apparent elimination half-life of carbamazepine, due to inhibition of its conversion to the epoxide, usually results in central nervous system (CNS) disturbances. Nightmares are troublesome in some patients. Allergic effects occur in about 0.5% of patients.
The estolate is particularly prone to give rise to liver abnormalities, consisting of upper abdominal pain, fever, hepatic enlargement, a raised serum bilirubin, pale stools and dark urine and eosinophilia. The condition is rare and usually seen 10–20 days after the initiation of treatment, with complete recovery on stopping the drug. Recurrence of symptoms can be induced by giving the estolate but not the base or stearate. There is evidence that erythromycin estolate is more toxic to isolated liver cells than is the 2′-propionate or the base, and it is suggested that the essential molecular feature responsible for toxicity is the propionyl–ester linkage. The relative frequency of the reaction, its rapidity of onset (within hours) after second courses of the drug, evidence of hypersensitivity and the histological appearance suggest a mixture of hepatic cholestasis, liver cell necrosis and hypersensitivity. Abnormal liver function tests in patients receiving the estolate must be interpreted with caution, since increased levels of transaminases is often the only abnormality and some metabolites of the estolate can interfere with the measurement commonly used. Elevated
levels of transaminases return to normal after cessation of treatment. Serum bilirubin is generally unchanged in these patients, but γ-glutamyl transpeptidase may also be affected. | | Safety Profile | Poison by intravenous
and intramuscular routes. Moderately toxic by ingestion, intraperitoneal, and
subcutaneous routes. An experimental
teratogen. Other experimental reproductive
effects. Mutation data reported. When
heated to decomposition it emits toxic
fumes of NOx. | | Synthesis | Erythromycin, (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-4-[(2,6-dideoxy-3-C�methyl-3-O-methyl-|á-L-ribo-hexopyranosyl)-oxy]-14-ethyl-7,12,13-trihydroxy-
3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(dimethylamino)-|?-D-xylo-hexopyranosyl]oxy
]oxacyclotetradecan-2,10-dione (32.2.1), is more specifically called erythromycin A. It was
first isolated in 1952 from the culture liquid of microorganisms of the type Streptomyces
erytherus. Minor amounts of erythromycin B and C were also found in the culture fluid.
Erythromycin B differs from A in that a hydrogen atom is located at position 12 in the place
of a hydroxyl group, while erythromycin C differs from A in that the residue of a different car�bohydrate, micarose (2-6-di-deoxy-3-C-methyl-L-ribohexose), is bound to the macrocycle in
position 3 in the place of cladinose (4-methoxy-2,4-dimethyl-tetrahydropyran-3,6-diol).
Erythromycin A is produced only microbiologically using active strains of microorgan�isms of the type Saccharopolospora erythraea.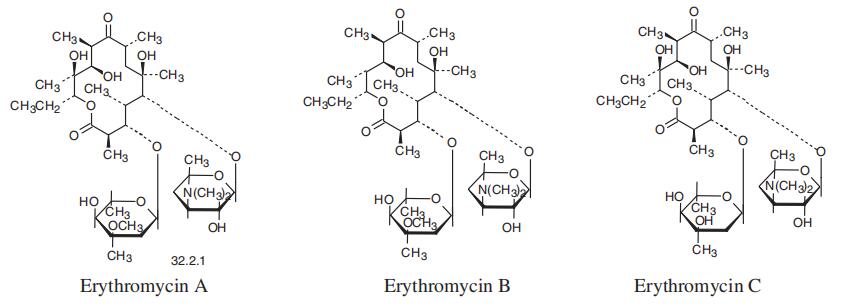
| | Veterinary Drugs and Treatments | Erythromycin is approved for use to treat infections caused by susceptible
organisms
in swine, sheep, and cattle. It is often employed
when an animal is hypersensitive to penicillins or if other antibiotics
are ineffective against a certain organism.
Erythromycin, at present, is considered to be one of the treatments
of choice (with rifampin) for the treatment of C. (Rhodococcus) equi
infections in foals. Erythromycin estolate and microencapsulated
base appear to be the most efficacious forms of the drug in foals due
to better absorption and less frequent adverse effects.
Erythromycin may be used as a prokinetic agent to increase gastric
emptying in dogs and cats. It may also be beneficial in treating
reflux esophagitis. | | Drug interactions | Potentially hazardous interactions with other drugsAminophylline and theophylline: inhibits
aminophylline and theophylline metabolism; if
erythromycin given orally decreased erythromycin
concentration.Anti-arrhythmics: increased risk of ventricular
arrhythmias with IV erythromycin and amiodarone
- avoid; increased toxicity with disopyramide;
increased risk of ventricular arrhythmias with
dronedarone - avoid.Antibacterials: increased risk of ventricular
arrhythmias with moxifloxacin and IV erythromycin
- avoid; possibly increased rifabutin concentration -
reduce rifabutin dose; concentration of bedaquiline
possibly increased - avoid if for more than 14 days;
possibly increased risk of ventricular arrhythmias
with delamanid; avoid with fidaxomicin.Anticoagulants: enhanced effect of coumarins;
concentration of edoxaban increased - reduce
edoxaban dose.Antidepressants: avoid concomitant use with
reboxetine; avoid IV erythromycin with citalopram
and escitalopram, risk of ventricular arrhythmias;
risk of ventricular arrhythmias with venlafaxine -
avoid.Antiepileptics: increased carbamazepine
concentration and possibly valproate.Antifungals: avoid with fluconazole.Antihistamines: possibly increases loratadine
concentration; inhibits mizolastine metabolism -
avoid concomitant use; concentration of rupatadine
increased.Antimalarials: avoid with artemether/lumefantrine;
increased risk of ventricular arrhythmias with
piperaquine with artenimol - avoid.Antimuscarinics: avoid concomitant use with
tolterodine.Antipsychotics: increased risk of ventricular
arrhythmias with sulpiride and zuclopenthixol
and IV erythromycin avoid; possibly increases
clozapine concentration leading to increased risk
of convulsions; possibly increased lurasidone
concentration; possibly increased risk of ventricular
arrhythmias with amisulpride, droperidol and
pimozide - avoid; possibly increased quetiapine
concentration Antivirals: concentration of both drugs increased
with telaprevir and simeprevir, avoid with simeprevir;
concentration increased by ritonavir; avoid with rilpivirine, concentration increased; increased risk of
ventricular arrhythmias with saquinavir - avoid. Anxiolytics and hypnotics: inhibits midazolam
and zopiclone metabolism; increases buspirone
concentration.Atomoxetine: increased risk of ventricular
arrhythmias with parenteral erythromycin.Avanafil: concentration of avanafil increased, max
dose 100 mg every 48 hours.Calcium-channel blockers: possibly inhibit
metabolism of calcium channel blockers; avoid with
lercanidipine.Ciclosporin: markedly elevated ciclosporin blood
levels - decreased levels on withdrawing drug.
Monitor blood levels of ciclosporin carefully and
adjust dose promptly as necessary.Cilostazol: concentration of cilostazol increased,
reduce cilostazol to 50 mg twice daily.Clopidogrel: possibly reduced antiplatelet effectColchicine: increased risk of colchicine toxicity -
suspend or reduce dose of colchicine, avoid in hepatic
or renal impairment.Cytotoxics: possibly increased afatinib concentration,
separate administration by 6-12 hours;
concentration of axitinib increased - reduce axitinib
dose; concentration of bosutinib possibly increased
- avoid or reduce dose of bosutinib; concentration
of cabozantinib, dasatinib and ibrutinib and
possibly olaparib increased - avoid with dasatinib,
reduce dose of ibrutinib, avoid or reduce dose of
olaparib; concentration of everolimus possibly
increased; increased risk of ventricular arrhythmias
with IV erythromycin and vandetanib - avoid;
possible interaction with docetaxel; increased risk
of ventricular arrhythmias with arsenic trioxide;
increases vinblastine toxicity - avoid.Diuretics: increased eplerenone concentration -
reduce eplerenone dose.Domperidone: possible increased risk of ventricular
arrhythmias - avoid.Ergot alkaloids: increase risk of ergotism - avoid
concomitant use.5HT1
agonists: increased eletriptan concentration -
avoid concomitant use.Ivabradine: increased risk of ventricular arrhythmias
- avoid concomitant use.Ivacaftor: concentration of ivacaftor possibly
increasedLipid-lowering drugs: possibly increased myopathy
with atorvastatin; concentration of pravastatin
increased; concentration of rosuvastatin reduced;
avoid concomitant use with simvastatin.1
;
concentration of lomitapide possibly increased -
avoid.Pentamidine: increased risk of ventricular
arrhythmias with IV erythromycin.Sildenafil: concentration of sildenafil increased -
reduce initial dose for ED or reduce frequency to
twice daily for PAH.Sirolimus: concentration of both drugs increased.Tacrolimus: markedly elevated tacrolimus blood
levels - decreased levels on withdrawing drug.
Monitor blood levels of tacrolimus carefully and
adjust dose promptly as necessaryTicagrelor: concentration of ticagrelor possibly
increased. | | Metabolism | Erythromycin is partly metabolised in the liver
by the cytochrome P450 isoenzyme CYP3A4 via
N-demethylation to inactive, unidentified metabolites.
It is excreted in high concentrations in the bile and
undergoes intestinal reabsorption. About 2-5% of an
oral dose is excreted unchanged in the urine and as much
as 12-15% of an intravenous dose may be excreted
unchanged by the urinary route. | | Purification Methods | It recrystallises from H2O to form hydrated crystals which melt at ca 135-140o, resolidifies and melts again at 190-193o. The melting point after drying at 56o/8mm is that of the anhydrous material and is at 137-140o. Its solubility in H2O is ~2mg/mL. The hydrochloride has m 170o, 173o (from aqueous EtOH, EtOH/Et2O). [Flynn et al. J Am Chem Soc 76 3121 1954, constitution: Wiley et al. J Am Chem Soc 79 6062 1957]. [Beilstein 18/10 V 398.] | | Dosage forms | 1 g/day in divided doses. |
| | Erythromycin Preparation Products And Raw materials |
|