4-bromo-2,1,3-benzothiadiazole manufacturers
|
| | 4-bromo-2,1,3-benzothiadiazole Basic information |
| Product Name: | 4-bromo-2,1,3-benzothiadiazole | | Synonyms: | IFLAB-BB F1918-0061;4-BROMO-2,1,3-BENZOTHIADIAZOLE;AKOS BBS-00002903;4-BROMO-2,1,3-BENZOTHIAZOLE;4-BroMobenzo[c][1,2,5]thiadiazole;4-Bromo-benzo[1,2,5]thiadiazole;4-Bromobenzo[2,1,3]thiadiazole;BT-BR | | CAS: | 22034-13-5 | | MF: | C6H3BrN2S | | MW: | 215.07 | | EINECS: | 1533716-785-6 | | Product Categories: | Pyridines;Halides;Oxadiazoles & Thiadiazoles;Oxadiazoles & Thiadiazoles | | Mol File: | 22034-13-5.mol | 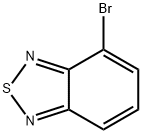 |
| | 4-bromo-2,1,3-benzothiadiazole Chemical Properties |
| Melting point | 80 °C | | Boiling point | 272.2±13.0 °C(Predicted) | | density | 1.859±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | pka | -1.02±0.45(Predicted) |
| Hazard Codes | Xi,Xn | | Risk Statements | 22 | | HS Code | 2934999090 |
| | 4-bromo-2,1,3-benzothiadiazole Usage And Synthesis |
| Uses | The Sonogashira cross-coupling reaction between 4-bromo-2, 1,3-benzothiadiazole, and the respective para-substituted phenylethynyl derivatives could synthesize the BTDs. In addition, the BTD-Ph is prepared by reacting 4-bromo-2,1,3-benzothiadiazole (Br-BTD)and palladium-MAI catalyst, and the dynamic phenylboronic acid reagent in the starting system or dynamic o-amino pyridine reagent[1-2].
| | References | [1] Pradhan, Asit Kumar et al. “Effects of donor and acceptor substituents on the photophysics of 4-ethynyl-2,1,3-benzothiadiazole derivatives?.” Physical Chemistry Chemical Physics 42 (2023): 29327–29340.
[2] AMARO DA SILVEIRA NETO B; CORREA J R; OLIVEIRA CARVALHO T; PIMENTA ROCHA CARVALHO P. "Selective cellular markers in selective cellular imaging of organelles such as mitochondria, nucleus and other organelles, are derived from 4-bromo-2,1,3-benzothiadiazole nucleus via homogeneous catalysis of palladium complex" BR102016025541(A2) |
| | 4-bromo-2,1,3-benzothiadiazole Preparation Products And Raw materials |
|