- (1R)-endo-(+)-Fenchol
-

- $0.00 / 100G
-
2020-02-26
- CAS: 2217-02-9
- Min. Order: 100G
- Purity: 99.0%+
- Supply Ability: 100 tons
|
| | (+)-Fenchol Basic information |
| Product Name: | (+)-Fenchol | | Synonyms: | FEMA 2480;ALPHA FENCHOL;(1R)-1,3,3-TRIMETHYLBICYCLO[2.2.1]HEPTAN-2-OL;(1R)-ENDO-(+)-FENCHOL;(1R)-ENDO-(+)-FENCHYL ALCOHOL;(+)-Fenchol~1,3,3-Trimethyl-2-norbornanol;1,3,3-TRIMETHYL-2-NORBORNANOL;D-Fenchol. | | CAS: | 2217-02-9 | | MF: | C10H18O | | MW: | 154.25 | | EINECS: | 606-948-7 | | Product Categories: | Miscellaneous | | Mol File: | 2217-02-9.mol | 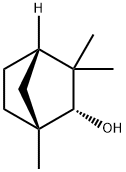 |
| | (+)-Fenchol Chemical Properties |
| Melting point | 43-46 °C | | Boiling point | 201-202 °C(lit.) | | density | 0.8389 (rough estimate) | | vapor pressure | 24.92-44.99Pa at 20-25℃ | | refractive index | 1.4790 (estimate) | | Fp | 165 °F | | storage temp. | 2-8°C | | solubility | Chloroform (Slightly), Ethanol (Slightly), Methanol (Slightly) | | form | Solid crystalline mass | | pka | 15.38±0.60(Predicted) | | color | colorless, translucent, occasionally pale straw-colored | | Odor | Lime-like odor with more or less of an earthy-dry character | | Odor Type | floral | | optical activity | [α]20/D +11.0±0.5°, c = 10% in ethanol | | Water Solubility | Insoluble in water. | | JECFA Number | 1397 | | BRN | 1850917 | | LogP | 2.93 at 20℃ and pH6.37-6.5 | | CAS DataBase Reference | 2217-02-9(CAS DataBase Reference) |
| Risk Statements | 36/38 | | Safety Statements | 22-24/25 | | RIDADR | UN 1325 4.1/PG 2 | | WGK Germany | 3 | | TSCA | Yes | | HS Code | 29061900 |
| | (+)-Fenchol Usage And Synthesis |
| Chemical Properties | clear slightly yellow liquid after melting | | Uses | (1R)-endo-(+)-Fenchyl alcohol?can be extensively used in daily chemicals such as fair antiperspirant, textile softener and liquid detergent. It can also be esterified by different kinds of organic acids to expand its usage in flavors and fragrances. | | Uses | Solvent, organic intermediate, odorant, flavoring. | | Preparation | By reduction of Fenchone, which
may be derived from Cedarleaf oil or from
other natural sources, or prepared synthetically from Pine terpenes. One French manufacturer uses Hydration of Turpentine under acid
conditions to produce the alcohol. | | General Description | (1R)-endo-(+)-Fenchyl alcohol is a bicyclic monoterpenoid that contains a fenchane skeleton. It is a commonly used volatile compound in fragrances and flavoring agents. |
| | (+)-Fenchol Preparation Products And Raw materials |
|