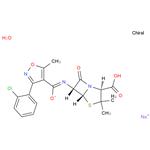
- Cloxacillin sodium
-
- $0.00 / 1kg
-
2023-11-01
- CAS:7081-44-9
- Min. Order: 1kg
- Purity: 99.0%
- Supply Ability: 20 tons
|
| | Sodium cloxacillin monohydrate Chemical Properties |
| Melting point | 170℃ | | Boiling point | 689℃ | | Fp | >110°(230°F) | | storage temp. | Keep in dark place,Inert atmosphere,2-8°C | | solubility | H2O: soluble50mg/mL | | form | solid | | color | White to Orange to Green | | Water Solubility | Soluble in water | | Merck | 13,2444 | | BRN | 5403885 | | CAS DataBase Reference | 7081-44-9(CAS DataBase Reference) |
| Hazard Codes | Xn | | Risk Statements | 36/37/38-42/43 | | Safety Statements | 26-36 | | WGK Germany | 2 | | RTECS | XH8920000 | | HS Code | 29411099 |
| | Sodium cloxacillin monohydrate Usage And Synthesis |
| Brand Name(s) in US | Cloxapen, Orbenin, Tegopen
| | Description | Cloxacillin was synthesized by Beecham Research Laboratories in 1962 starting with 6- aminopenicillanic acid. It was the first semisynthetic penicillin with a halogen atom in the side chain to be used clinically. It shows 5- to 20-fold stronger activity than methicillin and is twice as active as oxacillin against Staphylococcus aureus, including benzylpenicillin-resistant strains. Cloxacillin is highly stable against penicillinase and well absorbed by oral administration. It has been widely used, by oral and intramuscular administration, to treat internal, surgical, gynecological, and other infections caused by both benzylpenicillin-sensitive and benzylpenicillin-resistant bacteria. | | Chemical Properties | White or almost white, hygroscopic, crystalline powder. | | Uses | Penicillin antibacterial. | | Uses | Targets primarily Gram-positive bacteria, especially -lactamase producing Staphylococci | | Definition | ChEBI: Cloxacillin sodium monohydrate is an organic sodium salt and a hydrate. It contains a cloxacillin sodium. | | Brand name | Cloxapen (GlaxoSmithKline); Tegopen
(Apothecon). | | Contact allergens | Cloxacillin is a semisynthetic penicillin close to oxacillin.
It induced contact dermatitis in a pharmaceutical
factory worker with positive reactions to ampicillin,
but not to penicillin. In cutaneous drug reactions such
as acute generalized exanthematous pustulosis due to
amoxicillin, cross-reactivity is frequent to cloxacillin
(personal observations). | | Clinical Use | The chlorine atom ortho to the position of attachment of thephenyl ring to the isoxazole ring enhances the activity ofcloxacillin sodium, [3-(o-chlorophenyl)-5-methyl-4-isoxazolyl]penicillin sodium monohydrate (Tegopen), over that ofoxacillin, not by increasing its intrinsic antibacterial activitybut by enhancing its oral absorption, leading to higher plasmalevels. In almost all other respects, it resembles oxacillin. | | Safety Profile | Moderately toxic by intraperitoneal, intramuscular, subcutaneous, and intravenous routes. Mildly toxic by ingestion. When heated to decomposition it emits very toxic fumes of Cl-, NOx, Na2O, and SOx. | | Veterinary Drugs and Treatments | Cloxacillin is used via intramammary infusion in dry and lactating
dairy cattle. |
| | Sodium cloxacillin monohydrate Preparation Products And Raw materials |
|