|
|
| | 2-AMINO-1-PROPENE-1,1,3-TRICARBONITRILE Basic information |
| Product Name: | 2-AMINO-1-PROPENE-1,1,3-TRICARBONITRILE | | Synonyms: | 1,1,3-Tricyano-2-aminopropene;2-AMino-1-propene-1,1,3-tricarbonitrile, 97% 5GR;2-Amino-1,1,3-propenetricarbonitrile 97%;2-AMINO-1-PROPENE-1,1,3-TRICARBONITRILE;2-AMINO-1,1,3-TRICYANOPROPENE;2-Amino-1,1,3-trycino-1-propene;Aminotrycinopropene;3-aminoprop-1-ene-1,1,3-tricarbonitrile | | CAS: | 868-54-2 | | MF: | C6H4N4 | | MW: | 132.12 | | EINECS: | 212-777-5 | | Product Categories: | | | Mol File: | 868-54-2.mol | 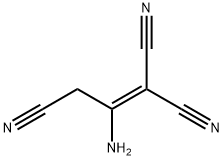 |
| | 2-AMINO-1-PROPENE-1,1,3-TRICARBONITRILE Chemical Properties |
| Melting point | 171-173 °C (lit.) | | Boiling point | 234.02°C (rough estimate) | | density | 1.262 | | refractive index | 1.7000 (estimate) | | storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | form | powder to crystal | | pka | -2.92±0.70(Predicted) | | color | White to Gray to Brown | | Merck | 14,480 | | BRN | 1756131 | | CAS DataBase Reference | 868-54-2(CAS DataBase Reference) |
| Hazard Codes | Xn | | Risk Statements | 22 | | Safety Statements | 22-24/25 | | RIDADR | UN 3259 8/PG 3 | | WGK Germany | 3 | | RTECS | UD2340000 | | HazardClass | 6.1 | | PackingGroup | III | | HS Code | 29269095 |
| | 2-AMINO-1-PROPENE-1,1,3-TRICARBONITRILE Usage And Synthesis |
| Chemical Properties | light pink to beige or light brownish. 2-Amino-1-propene-1,1,3-tricarbonitrile [868-54-2], 2-amino-1,1,3-tricyanopropene, malononitrile dimer, (NC)2C=C (NH2) CH2CN, Mr 132.13, mp 171 – 173℃, is a light brownish powder, insoluble in water.
| | Uses | 2-Amino-1,1,3-propenetricarbonitrile stimulates choline acetyltransferase activity in animals. A neurotrophic agent with potential therapeutic effect on cholinergic disorders. | | Biochem/physiol Actions | 2-Amino-1,1,3-propenetricarbonitrile is a key intermediate in synthesis of various heterocyclic compounds. 2-Amino-1,1-3-tricyanopropene undergoes condensation reaction with aromatic nitroso compounds to form useful coloring agents. 2-Amino-1,1-3-tricyanopropene reacts with 1,3-dithiole-derived polyenals to form push–pull systems with (Z)-geometry around the newly formed carbon-carbon double bond. | | Synthesis | Malononitrile dimer is obtained from the base-catalyzed dimerization of malononitrile. It has been investigated as a potential precursor for new dyes but it has no current industrial significance. |
| | 2-AMINO-1-PROPENE-1,1,3-TRICARBONITRILE Preparation Products And Raw materials |
|