| Company Name: |
Energy Chemical
|
| Tel: |
021-58432009 400-005-6266 |
| Email: |
marketing@energy-chemical.com |
| Products Intro: |
Product Name:Amisometradine
CAS:550-28-7
Purity:NULL Package:100mg;1g;25mg;500mg;50mg;5mg Remarks:NULL
|
|
| | amisometradine Basic information |
| Product Name: | amisometradine | | Synonyms: | 6-Amino-3-methyl-1-(2-methyl-2-propenyl)pyrimidine-2,4(1H,3H)-dione;6-Amino-3-methyl-1-(2-methylallyl)uracil;Aminoisometradine;Rolicton;6-amino-3-methyl-1-(2-methylallyl)pyrimidine-2,4-quinone;6-amino-3-methyl-1-(2-methylprop-2-enyl)pyrimidine-2,4-dione;6-azanyl-3-methyl-1-(2-methylprop-2-enyl)pyrimidine-2,4-dione;amisometradine | | CAS: | 550-28-7 | | MF: | C9H13N3O2 | | MW: | 195.22 | | EINECS: | 208-980-3 | | Product Categories: | | | Mol File: | 550-28-7.mol | 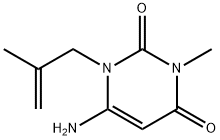 |
| | amisometradine Chemical Properties |
| Melting point | 175° | | Boiling point | 331.89°C (rough estimate) | | density | 1.1985 (rough estimate) | | refractive index | 1.5100 (estimate) | | storage temp. | Amber Vial, -20°C Freezer, Under inert atmosphere | | solubility | DMSO (Slightly), Methanol (Slightly) | | form | Solid | | pka | 4.97±0.70(Predicted) | | color | White to Pale Beige | | Stability: | Light Sensitive |
| | amisometradine Usage And Synthesis |
| Originator | Rolicton,Searle,US,1956 | | Uses | Amisometradine is an oral diuretic. | | Definition | ChEBI: Amisometradine is a pyrimidone. | | Manufacturing Process | Preparation of the ethyl analog is as follows (methyl isocyanate is used in
amisometradine manufacture).
To a cooled and stirred solution of 142 parts of methallylamine in 900 parts of
benzene, 156 parts of ethyl isocyanate are added dropwise. Upon
concentration in vacuum N-ethyl-N'-methallylurea is obtained.
260 parts of this urea derivative are dissolved in 500 parts of acetic anhydride
and treated with 157 parts of cyanoacetic acid at 60°C and heated at that
temperature for 2 hours. The solution is then concentrated in vacuum to a
syrup. 100 parts of water are added and the vacuum distillation is repeated.
The remaining syrup contains a mixture of N-cyanoacetyl-N-ethyl-N'-
methallylurea and a small quantity of N-cyanoacetyl-N-methallyl-N'-ethylurea.
This syrup is treated with sufficient 20% sodium hydroxide solution to raise
the pH to 10. A violent reaction occurs. The reaction mixture is diluted with 50
parts of water, stirred, cooled and filtered. The material collected on the filter
is recrystallized from 10% ethanol to yield a mixture of 1-methallyl-3-ethyl-6-
amino-1,2,3,4-tetrahydro-2,4-pyrimidinedione and 1-ethyl-3-methallyl-6-
amino-1,2,3,4-tetrahydro-2,4-pyrimidinedione melting at about 157-159°C. | | Therapeutic Function | Diuretic |
| | amisometradine Preparation Products And Raw materials |
|