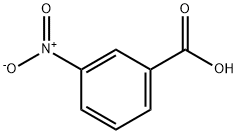
Reactivity and Polarity of 3-Nitrobenzoic acid
Dec 28,2023
3-Nitrobenzoic acid is an off-white solid, it is used to prepare some dyes.
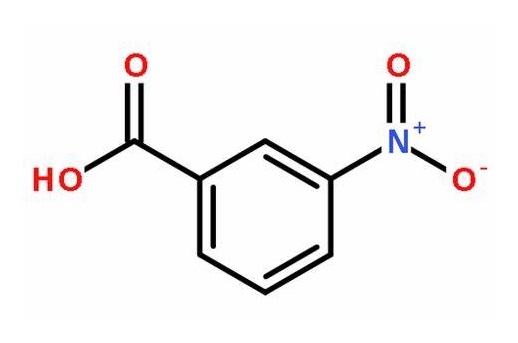
Properties
With a pKa of 3.47,3-nitrobenzoic acid is about ten times more acidic than benzoic acid. The conjugate base of benzoic acid is stabilized by the presence of the electron-withdrawing nitro group which explains its increased acidity in comparison to unsubstituted benzoic acid. It is typically soluble in oxygenated and chlorinated solvents.
Reactivity
The presence of both carboxylic acid and nitro functional groups deactivates the ring concerning electrophilic aromatic substitution reactions.
Polarity
3-Nitrobenzoic acid is a polar compound. This is because of the unbalanced electron density. The electronegativity difference between hydrogen and carbon is negligible, whereas the electronegativity difference between carbon and oxygen, nitrogen and oxygen is larger enough to cause polarity.
- Related articles
- Related Qustion
- Trypanocidal and anticorrosive effects of 3-Nitrobenzoic acid Dec 16, 2024
3-Nitrobenzoic acid is a commonly used chemical reagent, which is widely used as raw material for organic synthesis or intermediate for dyestuff and medicine.
Vanillyl alcohol ether derivatives, specifically vanillyl butyl ether, have been proposed as an alternative, milder warming agent for personal care applications.....
Dec 28,2023Biochemical EngineeringKojic acid is a small organic molecule with many potential applications, from pharmaceuticals to biopolymers and cosmetics.....
Dec 28,2023API3-Nitrobenzoic acid
121-92-6You may like
3-Nitrobenzoic acid manufacturers
- 3-Nitrobenzoic acid
-

- $15.35 / 25Kg/Drum
- 2025-10-28
- CAS:121-92-6
- Min. Order: 25Kg/Drum
- Purity: 99.50%HPLC
- Supply Ability: 10tons/month
- 3-Nitrobenzoic acid
-

- $10.00 / 1KG
- 2025-10-21
- CAS:121-92-6
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10 mt
- 3-Nitrobenzoic acid
-

- $100.00 / 1KG
- 2025-09-25
- CAS:121-92-6
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available