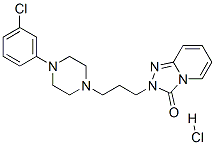
Trazodone Hydrochloride: From Absorption Kinetics to Therapeutic Efficacy in Major Depressive Disorder
Apr 2,2025
Trazodone hydrochloride is an antidepressant that belongs to a group of drugs called serotonin receptor antagonists and reuptake inhibitors (SARIs). This drug is used to treat major depressive disorder. It may help to improve your mood, appetite, and energy level as well as decrease anxiety and insomnia related to depression. Trazodone hydrochloride works by helping to restore the balance of a certain natural chemical (serotonin) in the brain.

Pharmacokinetics of single oral dose trazodone hydrochloride
Trazodone Hydrochloride is an antidepressant chemically unrelated to tri-cyclic, tetra-cyclic, or other known antidepressant agents. The mechanism of trazodone hydrochloride's antidepressant action in man is not fully understood. In animals, trazodone selectively inhibits its serotonin uptake by brain synaptosomes and potentiates the behavioral changes induced by the serotonin precursor, 5-hydroxytryptophan. Trazodone hydrochloride is not a monoamine oxidase inhibitor and, unlike amphetamine-type drugs, does not stimulate the central nervous system. Trazodone is rapidly and almost completely absorbed from the GI tract following oral administration. The rate and extent of absorption are affected by the presence of food. Peak plasma concentrations of trazodone occur approximately 1 h after oral administration when the drug is taken on an empty stomach or 2 h after oral administration when taken with food. When trazodone hydrochloride is taken shortly after the ingestion of food, there may be a slight increase (up to 20%) in the amount of drug absorbed.[1]
Distribution of trazodone hydrochloride into human body tissues and fluids has not been determined. Plasma concentrations of trazodone decline in a biphasic manner. Following oral administration of single doses of 25, 50, or 100 mg of trazodone to healthy, fasted adults in another study, mean peak plasma trazodone concentrations were 490, 860, and 1620 ng/mL, respectively. The half-life of trazodone in the initial phase is about 3–6 h and the half-life in the terminal phase is about 5–9 h. The clearance of trazodone hydrochloride from the body shows wide inter-individual variations. The therapeutic range for plasma trazodone concentrations and the relationship of plasma concentrations to clinical response and toxicity have not been established. The plasma samples of subjects were analyzed using a validated LC-MS/MS method for trazodone hydrochloride at the Bioanalytical facility of Lambda Therapeutic Research Ltd., Ahmedabad, India. The method was validated as per the regulatory guideline. Calibration curves using an eight-point calibration curve standards for trazodone, with concentration ranging from 5.2 ng/mL to 3025.2 ng/mL, were used to determine the concentration of trazodone hydrochloride in the samples of various subjects.
For the test formulation, the trazodone hydrochloride gMean Cmax was 1480.9 ng/mL (vs. 1520.2 ng/mL for reference) which is comparable to the published data 1470 ng/mL (Nilsen and Dale, 1992) where as in another study it was 1188 ng/mL after 300 mg extended release tablet (Jessica and Alan Caspi, 2011). In this study AUC0−t was 18193.0 ng·h/mL (vs. 18209.8 ng·h/mL) and AUC0−∞ was 19346.3 ng·h/mL (vs. 19393.4 ng·h/mL). In another study the same drug was tested under fasting conditions. For the test formulation, the trazodone hydrochloride gMean Cmax was 2172.2 ng/mL (vs. 2031.2 ng/mL for reference), AUC0−t was 16631.6 ng·h/mL (vs. 16342.9 ng·h/mL) and AUC0−∞ was 17460.6 ng·h/mL (vs. 17270.1 ng·h/mL).
Trazodone hydrochloride extended-release tablets
Trazodone hydrochloride is a once-daily, controlled-release formulation of trazodone indicated for the treatment of MDD in adults. Trazodone hydrochloride utilizes Contramid drug-delivery technology to control the release of trazodone over 24 hours (Karhu 2010). The pharmacokinetics of OLEPTRO are linear at doses ranging from 75 mg to 375 mg. OLEPTRO’s™ recommended starting dose is 150 mg and the maximum dose is 375 mg. Trazodone hydrochloride is not an AB-rated generic formulation of immediate-release trazodone. The Contramid technology distinguishes OLEPTRO from immediate-release trazodone. Contramid is a cross-linked modified high amylose starch matrix. Molecules of trazodone are embedded in this matrix. When exposed to fluid, a semi-permeable membrane forms, which provides the controlled-release mechanism of Trazodone hydrochloride and consistent release of trazodone over 24 hours, with a smooth pharmacokinetic curve versus peaks and troughs seen with immediate-release trazodone. The scored tables may be broken in half without compromising the controlled-release properties.[2]
Trazodone hydrochloride is not recommended for use during the initial recovery phase of myocardial infarction. Caution should be used when administering OLEPTRO to patients with cardiac disease and such patients should be closely monitored, since antidepressant drugs (including trazodone hydrochloride) may cause cardiac arrhythmias. QT prolongation has been reported with trazodone therapy. Clinical studies in patients with pre-existing cardiac disease indicate that trazodone hydrochloride may be arrhythmogenic in some patients in that population. Arrhythmias identified include isolated PVCs, ventricular couplets, tachycardia with syncope, and Torsades de Pointes. Postmarketing events have been reported at doses of 100 mg or less with the immediate-release form of trazodone.
OLEPTRO is a once-daily formulation of trazodone that utilizes Contramid drug-delivery technology to control the release of trazodone over 24 hours. This allows for consistent drug concentrations over 24 hours. OLEPTRO is available in bisectable tablets containing 150 mg or 300 mg of trazodone hydrochloride. Breaking the tablet in half does not affect the controlled-release properties of the tablet; however, to maintain its controlled-release properties, OLEPTRO should not be chewed or crushed. OLEPTRO is not an AB-rated generic formulation of immediate-release trazodone. Trazodone hydrochloride offers patients the benefits of a therapeutic antidepressant dose of trazodone and improvement in overall sleep quality when sleep disturbance is a symptom of depression. OLEPTRO™ is indicated for MDD in adults but is not indicated for insomnia.
Role of trazodone hydrochloride in treatment of major depressive disorder
Major depressive disorder (MDD) is the most common mood disorder and a leading cause of disability worldwide. Trazodone hydrochloride, a triazolopyridine serotonin receptor antagonist and reuptake inhibitor (SARI) antidepressant approved for major depressive disorder (MDD) in adults, has established efficacy that is comparable to other available antidepressants, and is effective for a range of depression symptoms, including insomnia, which is one of the most common and bothersome symptoms of depression. Also, Trazodone hydrochloride’s pharmacodynamic properties allow it to avoid the side effects of insomnia, anxiety and sexual dysfunction often associated with selective serotonin reuptake inhibitor antidepressants. In this narrative review, we have summarized recent clinical trials and real-world data on trazodone hydrochloride, including the recently introduced once-daily formulation, which has single dose pharmacokinetic properties that maintain effective blood trazodone hydrochloride levels for 24 h, while avoiding concentration peaks associated with side effects.[3]
This, combined with a low incidence of weight gain, and sexual dysfunction, may improve adherence to treatment. The most common adverse effects of trazodone hydrochloride are somnolence, headache, dizziness and xerostomia. It has minimal anticholinergic activity but may be associated infrequently with orthostatic hypotension (especially in patients with cardiovascular disease or older adults), QT interval prolongation, cardiac arrhythmias, and rare episodes of priapism. The low liability for activating side effects, the efficacy on symptoms such as insomnia and psychomotor agitation and the rapid onset of action make it useful for many depressed patients, both in monotherapy at nominal dosages of 150–300 mg/day, and in combination with other antidepressants at lower dosages. Appropriate doses for achieving antidepressant effect in adult patients are usually 150–300 mg/day and are often higher than the doses used when trazodone hydrochloride is prescribed to enhance the antidepressant effect of another drug (e.g., to treat insomnia). Trazodone hydrochloride is usually well tolerated and carries a low risk of anticholinergic side effects (e.g., constipation, urinary retention, xerostomia), weight gain, and sexual side effects.
References
[1]Kale P, Agrawal YK. Pharmacokinetics of single oral dose trazodone: a randomized, two-period, cross-over trial in healthy, adult, human volunteers under fed condition. Front Pharmacol. 2015 Oct 2;6:224.
[2]Oleptro™ (trazodone hydrochloride) extended-release tablets. P T. 2011 Feb;36(2):2-18.
[3]Fagiolini A, González-Pinto A, Miskowiak KW, Morgado P, Young AH, Vieta E. Role of trazodone in treatment of major depressive disorder: an update. Ann Gen Psychiatry. 2023 Sep 2;22(1):32.
- Related articles
- Related Qustion
Supplementation with pyridoxal 5'-phosphate monohydrate can synthesize neurotransmitters such as dopamine and serotonin, maintaining a healthy nervous system.....
Nov 4,2025Biochemical EngineeringVecuronium bromide is used to relax the muscles which works by blocking the signals between your nerves and your muscles.....
Apr 3,2025APITrazodone hydrochloride
25332-39-2You may like
Trazodone hydrochloride manufacturers
- Trazodone hydrochloride
-

- $0.00 / 100g
- 2025-12-19
- CAS:25332-39-2
- Min. Order: 100g
- Purity: 98.0%~102.0%
- Supply Ability: 100kg/month
- Trazodone hydrochloride
-

- $5.00/ KG
- 2025-12-19
- CAS:25332-39-2
- Min. Order: 1KG
- Purity: 99% hplc
- Supply Ability: 500TONS
- Trazodone hydrochloride
-

- $40.00 / 500mg
- 2025-12-16
- CAS:25332-39-2
- Min. Order:
- Purity: 99.52%
- Supply Ability: 10g