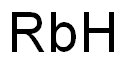What is the flame colour of rubidium?
Jan 26,2024
Rubidium is a soft, silvery-white metallic element of the alkali metals group (Group 1). It is one of the most electropositive and alkaline elements.
Rubidium can be liquid at ambient temperature, but only on a hot day given that its melting point is about 40°C. It ignites spontaneously in air and reacts violently with water and even with ice at -100 C, setting fire to the liberated hydrogen. As so with all the other alkali metals, it forms amalgams with mercury. It alloys with gold, cesium, sodium, and potassium. Its flame is yellowish-violet.

Applications
Rubidium and its salts have few commercial uses. The metal is used in the manufacture of photocells and in the removal of residual gases from vacuum tubes. Rubidium salts are used in glasses and ceramics and in fireworks to give them a purple colour. Potential uses are in ion engines for space vehicles, as working fluid in vapor turbines, and as getter in vacuum tubes.
Rubidium in the environment
Rubidium is considered to be the 16th most abundant element in the earth's crust. The relative abundance of rubidium has been reassessed in recent years and it is now suspected of being more plentiful than previously calculated. It is very like potassium and there are no environments where it is seen as a threat.
No minerals of rubidium are known, but rubidium is present in significant amounts in other minerals such as lepodite (1.5%), pollucite and carnallite. It is also present in traces in trace amounts in other minerals such as zinnwaldite and leucite.
The amount of rubidium produced every year is small, and what demand there is can be met from a stock of a mixed carbonate by-product that is collected during the extractium of litium from lepodite.
The little rubidium that is produced is used for research purposes only, these is no incentive to seek commercial outlets for the material.
- Related articles
- Related Qustion
- Application of Rubidium in Geology: Rb-Sr Isotope System May 28, 2024
This article will introduce the application of rubidium in geology, the Rb-Sr dating method, which is a radiometric dating technique utilized by geoscientists to determine the age of rocks and minerals.
- The Industrial Preparation of Rubidium Sep 26, 2019
Rubidium is a chemical element with the symbol Rb and atomic number 37. Rubidium is a very soft, silvery-white metal in the alkali metal group. Rubidium metal shares similarities to potassium metal and caesium metal in physical appearance,
- The applications of Rubidium Sep 26, 2019
Rubidium ,chemical symbol Rb, atomic number 37, and relative atomic mass (i.e., atomic weight) 85.4678(3), is the fourth alkali metal of group IA(1) of Mendeleev’s periodic chart. The word rubidium comes from the Latin rubidus, owing to the
Yes. The therapeutic fluoropyrimidines 5-fluorouracil (5-FU) and 5-fluorocytosine (5-FC) have long been used to treat human cancer and severe invasive fungal infections, respectively.....
Dec 16,2024Biochemical EngineeringThe synthetic route of Sotorasib is completed in three steps, starting with the amidation of nicotinic acid derivatives to produce nicotinamide, obtaining acyl isomer purity 28.6 by classical resolution, followed by the preparation of Borox....
Jan 26,2024InhibitorsRUBIDIUM
7440-17-7You may like