Polyacrylamide is a polymer formed from acrylamide subunits. Polyacrylamide has been extensively used in applications such as polyacrylamide gel electrophoresis.
Polyacrylamide resembles poly(acrylic acid) and poly(methacrylic acid)
in being water-soluble and, as with those polymers, it is mainly
this property which results in some limited commercial utilization.
Polyacrylamide is prepared by free radical polymerization, using techniques essentially similar to those described for poly(acrylic acid) and poly(methacrylic acid):
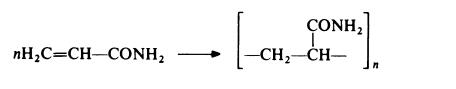
It may be noted that whereas this reaction gives a vinyl polymer, a different
type of polymer is obtained when polymerization is initiated by a strong base.
In this case, a polyamide (nylon 3) is formed. Active initiators for this type of
polymerization include alkoxides (RO-) and reaction probably takes place
according to the following scheme, which involves the rearrangement of a
carbanion to a more stable amide anion:
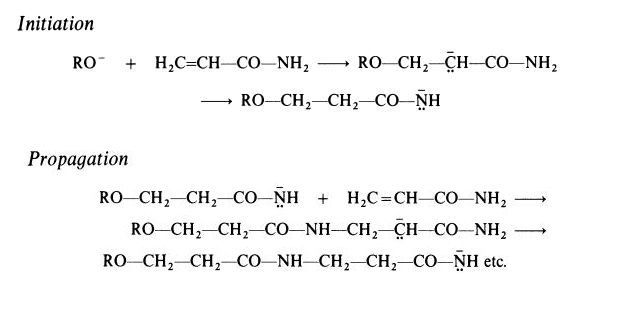
Polyacrylamide is a hard, brittle material. It is readily soluble in cold water
but solubility in organic compounds is generally very limited. The polymer
undergoes reactions characteristic of the amide group; for example, alkaline
hydrolysis introduces carboxylic groups and reaction with formaldehyde
gives methylol groups.
Polyacrylamide has found use as a ftocculant in the processing of minerals
and in water treatment. Copolymers of acrylamide and acrylic acid are used
to increase the dry strength of paper.
Intelligent Swelling/Collapsing copolymer that can be used as a temperature- and pH-sensitive material.