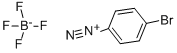1,1'-Biphenyl, 4'-bromo-2,4,6-trimethyl- synthesis
- Product Name:1,1'-Biphenyl, 4'-bromo-2,4,6-trimethyl-
- CAS Number:20434-38-2
- Molecular formula:C15H15Br
- Molecular Weight:275.18
Yield:20434-38-2 86%
Reaction Conditions:
with barium dihydroxide;tetrakis(triphenylphosphine) palladium(0) in 1,4-dioxane;water; for 22 h;Heating / reflux;
Steps:
20 Synthesis of 4-Bromo-2',4',6'-trimethylbiphenyl (GA35)
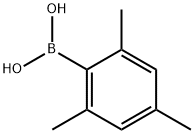
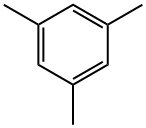
GA33 (4.98 mmol) obtained in Synthetic example 19, barium hydroxide octahydrate (14.9 mmol), Pd(PPh3)4 (0.24 mmol), degassed dioxane (33 mL), and distillated water (11 mL) were put in a three-necked round bottom flask and stirred. To the solution, GA31 (15.0 mmol) obtained in Synthetic example 18 was added, and the reaction mixture was heated and refluxed for 22 hours. The reaction mixture was cooled to room temperature. Dioxane was distilled away under a reduced pressure. After addition of 1 M hydrochloric acid, the mixture was extracted with dichloromethane three times. The combined extracts was washed with 1 M hydrochloric acid twice and with brine once, and dried over anhydrous sodium sulfate. After the drying, the extract was filtered. By means of column chromatography, the obtained solid was firstly separated with Hexane:CH2Cl2=2:1, and then separated and purified with Hexane:CH2Cl2=1:1 to give GA35 (11.50 mmol, yield: 86%). Rf=0.6. 1H-NMR(400MHz,CDCl3) δ = 7.52(d,2H,J=8.2Hz), 7.00(d,2H,J=8.2Hz), 6.92(s,2H), 2.31(s,3H), 1.98(s,6H). 13C-NMR(100MHz,CDCl3) δ = 139.95, 137.66, 136.89, 135.74, 131.56, 131.07, 128.13, 120.60, 20.99, 20.68.
References:
EP1623971,2006,A1 Location in patent:Page/Page column 20
576-83-0
246 suppliers
$27.00/25G

20434-38-2
3 suppliers
inquiry