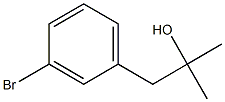
1-(3-Bromophenyl)-2-methylpropan-2-ol synthesis
- Product Name:1-(3-Bromophenyl)-2-methylpropan-2-ol
- CAS Number:3762-39-8
- Molecular formula:C10H13BrO
- Molecular Weight:229.11
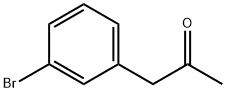
21906-32-1
98 suppliers
$40.00/500mg

75-16-1
265 suppliers
$12.00/10ml

3762-39-8
10 suppliers
inquiry
Yield:3762-39-8 83%
Reaction Conditions:
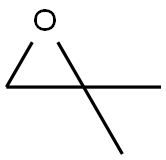
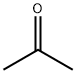
Stage #1: 3-bromophenylacetone;methylmagnesium bromide in diethyl ether at 0; for 3 h;
Stage #2: with ammonium chloride in diethyl ether;water at 0;
Steps:
35
Methylmagnesium bromide (3M solution in diethyl ether, 51.6 mL, 155 mmol) was slowly added to a solution of 1- (3-bromo-phenyl) propan-2-one (15.0 g, 70 mmol) in dry diethyl ether (200 mL) at 0 °C and the mixture was stirred for 3 hours. The reaction mixture was then re-cooled to 0 °C and slowly quenched with saturated aqueous ammonium chloride solution. The organic solution was washed with brine, dried over sodium sulfate and concentrated in vacuo. The residual yellow oil was then purified by column chromatography on silica gel eluting with dichloromethane : pentane: methanol, 90: 5: 5, to afford a pale-yellow oi . in83% yie . d, 13. 26 g. H NMR (400MHz, CDCI3) 8 : 1.22 (6H, s), 1.42 (1H, bs), 2. 74 (2H, s), 7.15 (2H, m), 7.40 (2H, m)
References:
WO2005/92840,2005,A1 Location in patent:Page/Page column 66
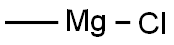
676-58-4
276 suppliers
$15.00/25ml
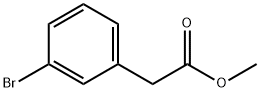
150529-73-0
151 suppliers
$6.00/1g

3762-39-8
10 suppliers
inquiry

21906-32-1
98 suppliers
$40.00/500mg

3762-39-8
10 suppliers
inquiry