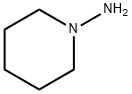
1-Aminopiperidine synthesis
- Product Name:1-Aminopiperidine
- CAS Number:2213-43-6
- Molecular formula:C5H12N2
- Molecular Weight:100.16
Yield:2213-43-6 74%
Reaction Conditions:
in methanol for 25 h;Reflux;
Steps:
N-aminopiperidine (2)
In a 100 ml two-necked flask, hydrazine hydrate (14.55 ml, 0.3 mol) was dissolved in 25 ml of methanol. Solution was heated to reflux and 1,5-dibromopentane (13.62 ml, 0.1 mol) was added dropwise within 1 h. Reaction mixture was left stirring at reflux for 24 h. Methanol was removed under reduced pressure and residue was basified with 40 g of 40% NaOH solution. Mixture was extracted 8 times with Et2O and combined extracts were dried on anhydrous Na2SO4 and filtrated. Solvent was removed under reduced pressure (40 C at 40 mbar). Process yielded 7.380 g (74%) of N-aminopiperidine, a light yellow liquid, which was used in subsequent experiments without further purification.
References:
Ilisson, Mihkel;Tomson, Kristjan;Selyutina, Anastasia;Türk, Silver;Mäeorg, Uno [Synthetic Communications,2015,vol. 45,# 11,p. 1367 - 1373] Location in patent:supporting information
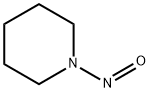
100-75-4
100 suppliers
$47.20/40458

2213-43-6
200 suppliers
$48.89/5g
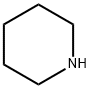
110-89-4
3 suppliers
$15.96/100ML

2213-43-6
200 suppliers
$48.89/5g
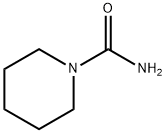
2158-03-4
31 suppliers
inquiry

2213-43-6
200 suppliers
$48.89/5g
![2,2,4,4-tetramethyl-7-oxa-3,20-diazadispiro[5.1.11.2]-henicosan-21-one](/CAS/GIF/64338-16-5.gif)
64338-16-5
5 suppliers
inquiry
584-08-7
1209 suppliers
$5.00/100g

106-95-6
429 suppliers
$10.00/5g

2213-43-6
200 suppliers
$48.89/5g

