
1-(2,2-difluoroethyl)-4-(tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole synthesis
- Product Name:1-(2,2-difluoroethyl)-4-(tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole
- CAS Number:1049730-40-6
- Molecular formula:C11H17BF2N2O2
- Molecular Weight:258.07

269410-08-4
391 suppliers
$5.00/1g
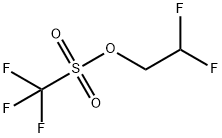
74427-22-8
169 suppliers
$18.00/1g

1049730-40-6
53 suppliers
$149.00/100mg
Yield: 36%
Reaction Conditions:
with caesium carbonate in N,N-dimethyl-formamide at 20; for 72 h;
Steps:
1-(2,2-Difluoroethyl)-4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)-1 H-pyrazole (I H-I 2)
2,2-Difluoroethyl trifluoromethanesulfonate (1 .00 g, 4.67 mmol) was added to a mixture of 4-(4,4,5,5-tetramethyl-1 ,3,2-dioxaborolan-2-yl)-1 H-pyrazole (0.91 mg, 4.67 mmol) and caesium carbonate (3.04 g, 9.34 mmol) in dry N,N-dimethylformamide (18 mL). The resulting mixture was stirred at room temperature for 72 h. The reaction mixture was diluted with ethyl acetate (150 mL) and washed with brine (3x100 mL). The organic layer was dried with sodium sulfate and concentrated in vacuo. Purification by flash column chromatography (Method L7; 12 g; heptane, 10%-30% ethyl acetate) afforded 0.44 g (1.68 mmol; 36% of theory) of the title compound.GO-MS (Method L9): R1 = 3.58 mm; mlz = 258 M1H NMR (300 MHz, Ohloroform-d, Method M2) 6 7.83 (s, 1H), 7.76 (s, 1H), 6.09 (tt, J = 55.5,4.3 Hz, 1H), 4.47 (td, J = 13.5, 4.3 Hz, 2H), 1.32 (s, 12H).
References:
BAYER ANIMAL HEALTH GMBH;KÖHLER, Adeline;WELZ, Claudia;BÖRNGEN, Kirsten;KULKE, Daniel;ILG, Thomas;KÖBBERLING, Johannes;HÜBSCH, Walter;SCHWARZ, Hans-Georg;GÖRGENS, Ulrich;EBBINGHAUS-KINTSCHER, Ulrich;HINK, Maike;NENNSTIEL, Dirk;RAMING, Klaus;ADAMCZEWSKI, Martin;BÖHM, Claudia WO2017/178416, 2017, A1 Location in patent:Page/Page column 113; 114
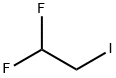
598-39-0
104 suppliers
$13.00/1g
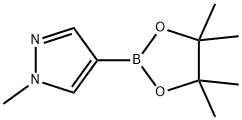
761446-44-0
355 suppliers
$10.00/1g

1049730-40-6
53 suppliers
$149.00/100mg

269410-08-4
391 suppliers
$5.00/1g

598-39-0
104 suppliers
$13.00/1g

1049730-40-6
53 suppliers
$149.00/100mg