(5-(ethylamino)-5-oxopentyl)triphenylphosphonium bromide synthesis
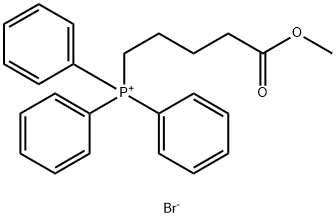
- Product Name:(5-(ethylamino)-5-oxopentyl)triphenylphosphonium bromide
- CAS Number:1201226-16-5
- Molecular formula:C25H29BrNOP
- Molecular Weight:470.3816
Yield:1201226-16-5 90%
Reaction Conditions:
with bromine in water at 25; for 60 h;
Steps:
1.2.2 Step 2: Preparation of compound of formula (VI)
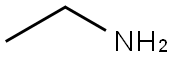
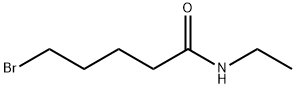
2) Preparation of (5- (ethylamino) -5-oxopentyl) triphenylphosphonium bromide 500 ml 3-neck flask The above-prepared (5-methoxy-5-oxopentyl) triphenylphosphonium Bromide into a (9.5 g, 0.021 mol, 1 eq) to 285 ml of ethyl amine (70% in water) And the mixture was stirred at 25 DEG C for 60 hours. The reaction was concentrated under reduced pressure. To the concentrate, 100 ml of H2O, 100 ml of methylene chloride was added and the layers were separated. The H2O layer was extracted one more time with 100 ml of methylene chloride, The organic layer was washed with 10 ml of a 10% aqueous NaCl solution. After drying with Na2SO4, the mixture was filtered and concentrated under reduced pressure to obtain an oil phase compound (8.9 g, 90%).
References:
KR2017/25682,2017,A Location in patent:Paragraph 0093; 0103; 0108-0110
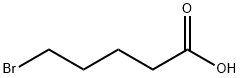
2067-33-6
376 suppliers
$6.00/5g

1201226-16-5
24 suppliers
inquiry
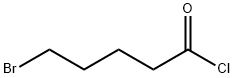
4509-90-4
227 suppliers
$15.00/1g

1201226-16-5
24 suppliers
inquiry
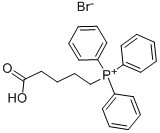
17814-85-6
388 suppliers
$8.00/5g

1201226-16-5
24 suppliers
inquiry