
1-Piperidinecarboxylic acid, 4-(2-propyn-1-yloxy)-, 1,1-dimethylethyl ester synthesis
- Product Name:1-Piperidinecarboxylic acid, 4-(2-propyn-1-yloxy)-, 1,1-dimethylethyl ester
- CAS Number:1219827-56-1
- Molecular formula:C13H21NO3
- Molecular Weight:239.31

106-96-7
339 suppliers
$31.00/25g
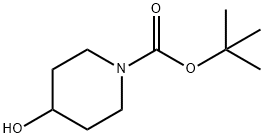
109384-19-2
469 suppliers
$5.00/1g

1219827-56-1
34 suppliers
inquiry
Yield:1219827-56-1 100%
Reaction Conditions:
Stage #1: 1-(tert-Butyloxycarbonyl)-4-piperidinolwith sodium hydride in tetrahydrofuran;mineral oil at 0; for 0.5 h;
Stage #2: propargyl bromide in tetrahydrofuran;mineral oil at 25; for 12 h;
Steps:
1 Step 1-Tert-butyl 4-prop-2-ynoxypiperidine-1-carboxylate (Intermediate)
To a solution of tert-butyl 4-hydroxypipenrdine-1-carboxylate 2.00 g, 9.94 mmol, CAS109384-19-2) in anhydrous THF (10 mL) was cooled to 0° C., and subsequently NaH (477 mg, 11.9 mmol, 60% oil dispersion) was added. The reaction mixture was stirred at 0° C. for 0.5 hr. Then, 3-bromoprop-1-yne (1.18 g, 9.94 mmol, 856 uL) was added. The resulting reaction mixture was stirred at 25° C. for 12 hrs. On completed, the reaction mixture was quenched with water (1 mL), then diluted with ethyl acetate (100 mL). The organic layers were washed with brine (20 mL), dried over anhydrous sodium sulfate, filtered and the filtrate was concentrated in vacuo. The residue was purified by column chromatography to give the title compound (2.38 g, 100% yield) as yellow oil. 1H NMR (400 MHz, CDCl3) δ 4.22 (d, J=2.4 Hz, 2H), 3.84-3.75 (m, 2H), 3.73-3.70 (m, 1H), 3.15-3.09 (m, 2H), 2.43 (t, J=2.4 Hz, 1H), 1.93-1.82 (m, 2H), 1.61-1.50 (m, 2H), 1.47 (s, 9H).
References:
US2019/192668,2019,A1 Location in patent:Paragraph 3869; 3870