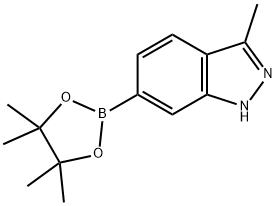
3-Methyl-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-indazole synthesis
- Product Name:3-Methyl-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-indazole
- CAS Number:1227911-51-4
- Molecular formula:C14H19BN2O2
- Molecular Weight:258.12
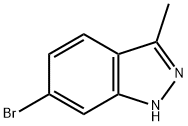
7746-27-2
117 suppliers
$12.00/250mg
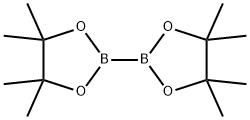
73183-34-3
559 suppliers
$6.00/5g

1227911-51-4
45 suppliers
$45.00/50mg
Yield:1227911-51-4 618 mg
Reaction Conditions:
with dichloro(1,1'-bis(diphenylphosphanyl)ferrocene)palladium(II)*CH2Cl2;potassium acetate in 1,4-dioxane at 150; for 0.5 h;Microwave irradiation;
Steps:
5.1 (1) Synthesis of 3-methyl-6-(4,4,5,5-tetramethyl[1,3,2]dloxaborolan-2-yl)-1H-indazole [5-1] (hereinafter referred to as a compound [5-1])
(1) Synthesis of 3-methyl-6-(4,4,5,5-tetramethyl[1,3,2]dloxaborolan-2-yl)-1H-indazole [5-1] (hereinafter referred to as a compound [5-1]) To a solution of 6-bromo-3-methyl-1H-indazole (647 mg), which was obtained by the method described in the document (JP 2009-528363 W), in 1,4-dioxane (10 mL) were added bis(pinacolato)diboron (1.15 g), potassium acetate (899 mg) and [1,1'-bis(diphenylphosphino)ferrocene]dichloropalladium(II) dichloromethane complex (126 mg), and the mixture was subjected to microwave irradiation at 150°C for 30min. The reaction mixture was quenched with water, and the mixture was extracted with ethyl acetate. The obtained organic layer was dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure. The obtained residue was purified by silica gel column chromatography to give the titled compound (618 mg) as a yellow solid. 1H-NMR (400 MHz, CDCl3) δ: 7.93 (1H, s), 7.67 (1H, dd, J = 8.1, 0.7 Hz), 7.55 (1H, d, J = 8.3 Hz), 2.60 (3H, s), 1.37 (12H, s).
References:
EP2878594,2015,A1 Location in patent:Paragraph 0401-0403
57848-46-1
384 suppliers
$9.00/1g

1227911-51-4
45 suppliers
$45.00/50mg
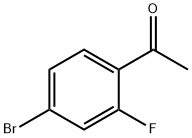
625446-22-2
192 suppliers
$6.00/250mg

1227911-51-4
45 suppliers
$45.00/50mg

353282-88-9
16 suppliers
inquiry

1227911-51-4
45 suppliers
$45.00/50mg