
N-(4-Bromobenzyl)-2-butyl-4-chloro-1H-imidazole-5-carboxyaldehyde synthesis
- Product Name:N-(4-Bromobenzyl)-2-butyl-4-chloro-1H-imidazole-5-carboxyaldehyde
- CAS Number:143722-29-6
- Molecular formula:C15H16BrClN2O
- Molecular Weight:355.66

83857-96-9
498 suppliers
$8.00/10g
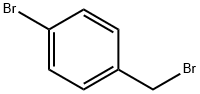
589-15-1
435 suppliers
$6.00/10g

143722-29-6
35 suppliers
$66.00/1g
Yield:143722-29-6 89.9%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl acetamide;water;toluene;
Steps:
14 2-n-Butyl-4-chloro-1-p-bromobenzyl-1H-imidazole-5-carboxaldehyde
EXAMPLE 14 2-n-Butyl-4-chloro-1-p-bromobenzyl-1H-imidazole-5-carboxaldehyde A mixture of 2-n-butyl-4-chloro-1H-imidazole-5-carboxaldehyde (0.6 m=111.9 g), p-bromobenzylbromide (0.6 m=153.02 g), anhydrous potassium carbonate (0.75 m=103.5 g), and dry N,N-dimethylacetamide (900 mL) was stirred at room temperature for 4 hours. The mixture was diluted with 1.2L of toluene and 1.8L of water. After mixing for half an hour, the layers were separated. The organic layer was washed two more times with 900 mL portions of water, then dried over magnesium sulfate. The drying agent was removed by filtration and the filtrate was concentrated. The residual oil was pumped overnight to a weight of 191.71 g (89.9% yield).
References:
US5310928,1994,A