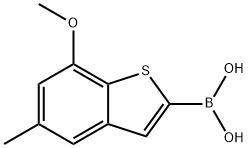
(7-Methoxy-5-Methylbenzo[b]thiophen-2-yl)boronic acid synthesis
- Product Name:(7-Methoxy-5-Methylbenzo[b]thiophen-2-yl)boronic acid
- CAS Number:1443531-60-9
- Molecular formula:C10H11BO3S
- Molecular Weight:222.07
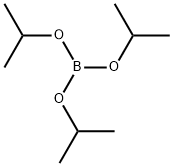
5419-55-6
371 suppliers
$12.00/5g
![7-Methoxy-5-Methylbenzo[b]thiophene](/CAS/20150408/GIF/1388033-29-1.gif)
1388033-29-1
24 suppliers
$155.00/50mg
![(7-Methoxy-5-Methylbenzo[b]thiophen-2-yl)boronic acid](/CAS/20150408/GIF/1443531-60-9.gif)
1443531-60-9
39 suppliers
$45.00/2.5mg
Yield:1443531-60-9 94%
Reaction Conditions:
Stage #1:7-methoxy-5-methyl-1-benzothiophene with n-butyllithium in tetrahydrofuran at -70; for 1 h;
Stage #2:Triisopropyl borate in tetrahydrofuran at -70; for 1 h;
Steps:
1.6 Step 6: Preparation of (7-methoxy-5-methylbenzothiophen-2-yl)boronic acid
1.5 g of 7-methoxy-5-methylbenzothiophene was dissolved in 20 ml of dry tetrahydrofuran, cooled to -70 °C, and then 1.5 equivalents of n-butyllithium solution was added dropwise, and after stirring at -70 °C for 1 hour, 3 equivalents of triisopropyl borate were added dropwise, and the mixture was stirred at -70 °C for 1 hour, then slowly warmed to room temperature. The reaction was quenched with saturated aqueous solution of ammonium chloride, stirred at room temperature for half an hour and then extracted with ethyl acetate. After washed with saturated aqueous solution of sodium chloride, the organic phase was dried over anhydrous sodium sulfate, filtered and concentrated, and the residue was separated by column chromatography (dichloromethane : methanol = 98:2) to afford 1.7 g of beige solids, yield 94%.
References:
Shanghai Institute of Materia Medica, Chinese Academy of Sciences;DUAN, Wenhu;GENG, Meiyu;WANG, Yuming;AI, Jing;FAN, Jun;DAI, Yang;DING, Jian EP3486244, 2019, A1 Location in patent:Paragraph 0123; 0124
![7-Methoxy-5-Methylbenzo[b]thiophene](/CAS/20150408/GIF/1388033-29-1.gif)
1388033-29-1
24 suppliers
$155.00/50mg
![(7-Methoxy-5-Methylbenzo[b]thiophen-2-yl)boronic acid](/CAS/20150408/GIF/1443531-60-9.gif)
1443531-60-9
39 suppliers
$45.00/2.5mg
80041-89-0
231 suppliers
$13.50/1G
![7-Methoxy-5-Methylbenzo[b]thiophene](/CAS/20150408/GIF/1388033-29-1.gif)
1388033-29-1
24 suppliers
$155.00/50mg
7732-18-5
462 suppliers
$12.69/100ml
![(7-Methoxy-5-Methylbenzo[b]thiophen-2-yl)boronic acid](/CAS/20150408/GIF/1443531-60-9.gif)
1443531-60-9
39 suppliers
$45.00/2.5mg

83485-34-1
3 suppliers
inquiry
![(7-Methoxy-5-Methylbenzo[b]thiophen-2-yl)boronic acid](/CAS/20150408/GIF/1443531-60-9.gif)
1443531-60-9
39 suppliers
$45.00/2.5mg
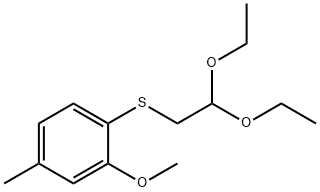
1443531-59-6
5 suppliers
inquiry
![(7-Methoxy-5-Methylbenzo[b]thiophen-2-yl)boronic acid](/CAS/20150408/GIF/1443531-60-9.gif)
1443531-60-9
39 suppliers
$45.00/2.5mg