
Toluene-4-sulfonic acid 4-tert-butoxycarbonylamino-butyl ester synthesis
- Product Name:Toluene-4-sulfonic acid 4-tert-butoxycarbonylamino-butyl ester
- CAS Number:180851-50-7
- Molecular formula:C16H25NO5S
- Molecular Weight:343.44
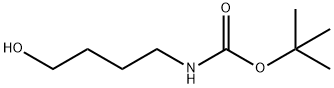
75178-87-9
119 suppliers
$6.00/100mg
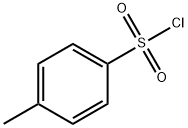
98-59-9
587 suppliers
$9.00/5g

180851-50-7
23 suppliers
$784.00/5g
Yield:180851-50-7 82.2%
Reaction Conditions:
with N,N-dimethyl-4-aminopyridine;triethylamine in dichloromethane at 0 - 20; for 7 h;
Steps:
33.1 Step 1: Preparation of 4-((tert-butoxycarbonyl)amino)butyl 4-methylbenzenesulfonate
To a stirred solution of tert-butyl N-(4-hydroxybutyl)carbamate (30.6 g, 162 mmol) in DCM (400 mL) was added DMAP (2.96 g, 24.2 mmol), TEA (40.9 g, 404 mmol) and p-toluenesulfonyl chloride (46.2 g, 242 mmol) at 0 °C. The resulting mixture was stirred for 2 h at 0 °C, then 5 h at room temperature. The solution was concentrated under reduced pressure and the residue was purified by silica gel column chromatography (petroleum ether/THF) to afford 4-((tert-butoxycarbonyl)amino)butyl 4-methylbenzenesulfonate (45.6 g, 82.2%) as a light-yellow oil. LCMS (ESI) m/z: [M+H]+ = 344.
References:
WO2022/109396,2022,A1 Location in patent:Page/Page column 64

24424-99-5
824 suppliers
$13.50/25G

180851-50-7
23 suppliers
$784.00/5g

75178-87-9
119 suppliers
$6.00/100mg

180851-50-7
23 suppliers
$784.00/5g